The Discovery of The Bacterium Behind 5% of All Cancers — Barry Marshall

One bacterium causes roughly 1 in 20 cancer cases worldwide. It’s the most cancer-causing pathogen we’ve found—and the main cause of peptic ulcers. Its discovery overturned an ironclad medical dogma that the stomach was sterile.
Despite infecting about half of humanity, Helicobacter pylori wasn't discovered until 1979 and shown to cause gastritis and peptic ulcer disease in the early 1980s. Why did it evade detection for so long—and what finally broke through the consensus?
I went to Perth, Australia—where H. pylori was first discovered—to chat with Barry Marshall, gastroenterologist and co-recipient (with Robin Warren) of the 2005 Nobel Prize in Physiology or Medicine for discovering H. pylori and proving that it causes gastritis and peptic ulcer disease. Marshall famously infected himself with the bacterium to demonstrate causality and later helped develop clinical diagnostics like the urea breath test, which we demo live in the episode.
We discuss:
- the rise and fall of stomach cancer in the West;
- whether Darwin’s dyspepsia and Napoleon's stomach cancer trace to H. pylori;
- the ulcer–cancer paradox;
- Correa’s cascade: what H. pylori eradication reverses—and what it doesn’t;
- the “H. pylori enigmas” (Africa, India, Costa Rica);
- eradication prospects and an oral vaccine timeline;
- how the field missed the discovery;
- how the primitive internet enabled the discovery;
- what the H. pylori discovery teaches us about how knowledge diffuses;
- lessons from manufacturing millions of tests in Perth;
- and much, much more.
Video
Sponsors
- Eucalyptus: the Aussie startup providing digital healthcare clinics to help patients around the world take control of their quality of life. Euc is looking to hire ambitious young Aussies and Brits. You can check out their open roles at eucalyptus.health/careers.
- Vanta: helps businesses automate security and compliance needs. For a limited time, get one thousand dollars off Vanta at vanta.com/joe. Use the discount code "JOE".
Transcript
JOSEPH WALKER: Today it’s my great honour to be speaking with Barry Marshall.
Barry is a gastroenterologist, professor emeritus of clinical microbiology, and along with Robin Warren he shared the 2005 Nobel Prize in Physiology or Medicine for their discovery of the bacterium Helicobacter pylori and its role in causing gastritis and peptic ulcer disease.
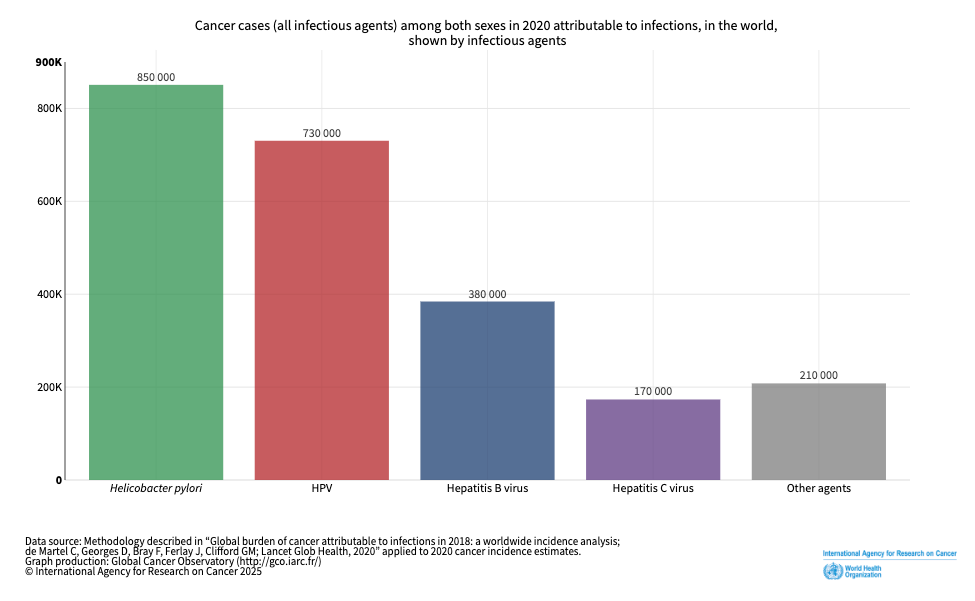
Just to put the scale of that in context as a humanitarian achievement, about 50% of the world’s human population is infected with H. pylori. It doesn’t just cause gastritis and peptic ulcers; it also causes gastric cancer (or stomach cancer).
Stomach cancer is the fifth most common cancer in the world. In recent years, it’s been the fourth or fifth most common cause of cancer deaths, killing about 800,000 people per year.
H. pylori is responsible for approximately 90% of non-cardia stomach cancer cases—that’s the more common form of stomach cancer. It can also cause some rare stomach cancers like MALT lymphoma.
That means H. pylori, this one bacterium, is responsible for about 5% of the total burden of all cancers globally—so one in 20 new cancers are caused by H. pylori. It’s the most cancer-causing pathogen that we know of, more so than even HPV or hepatitis B.
And thanks to Barry’s work, we’re now able to prevent these cancer cases through a simple eradication treatment of H. pylori.
So, Barry, welcome to the podcast.
BARRY MARSHALL: Thanks very much, Joe. Good introduction, and thank you for doing, obviously, a lot of homework on that.
WALKER: Of course, my pleasure. I’ve maybe spent about a week reading up on H. pylori and your work, and I’m going to try to unload as many of my questions on you now.
First question: it’s well documented that poor old Charles Darwin struggled with dyspepsia most of his adult life. He had stomach pain, nausea, and if you read his letters or his diary he complains about this a lot. He and his doctor put it down to nervous dyspepsia, so it also ruined his social life and turned him into a recluse.
How good is the evidence that it was actually H. pylori that was causing his problem?
MARSHALL: That is the subject of a rejected letter to Nature that a friend of mine submitted. I don’t know whether I was co-author, but I knew about it. The epidemiology is that in the 19th century the whole human race was infected with it, and there are many famous characters in the 19th century who died from stomach trouble or had it their whole life, which made them particularly cranky and intolerant.
Charles Darwin would have had it—most people had it—and he had this issue that whenever he was under stress or pressure, he would get these vomiting attacks. There were all kinds of theories about why he had “nervous dyspepsia”—the fact that he was in seminary school and later became an atheist, you see. He used to call himself the “devil’s chaplain” because he didn’t really believe it anymore. All the Freudians came up with reasons why he had stomach trouble and he was conflicted, et cetera.
But like anything that you don’t understand in medicine, and a lot of other areas, if we don’t know the cause of it, we say it’s caused by stress—because every person on earth has stress, so you can always blame it on the patients. Everyone said it was stress. In his biography, of course, even Alfred Nobel had the same diagnosis with stomach aches and so-called stress.
But Charles Darwin is an interesting one because, if you look at the details, his vomiting attacks started before his boat left Southampton on the voyage of the Beagle. On the voyage he supposedly got “seasickness” all the time, but you used to have to go and sit on the boat for a few days while they were waiting for the right tide and loading it up.
So he was in his cabin on the Beagle and had vomiting attacks then, even before he went on the voyage. So the story of it being seasickness is pretty much bogus.
On the voyage of the Beagle he had episodes where he would be laid up for days, and everyone said, “Oh, that was Chagas disease in South America,” and some other things.
For his whole life, he was a member of the Royal Society, but he couldn’t really travel up to London except once every 10 years to give a speech, because he would decompensate. He was always vomiting, he was always taking health cures like icy cold water and things like that. He even was treated on one occasion with bismuth treatment, which probably put him into remission because it does kill the H. pylori. So they snagged a good treatment at one point.
[Paragraph redacted as the paper Barry mentions is not yet published.]
So yes, he did have Helicobacter pylori, and that is certainly the most likely explanation for Charles Darwin’s lifelong guts aches.
WALKER: That’s amazing. This feels like a scoop, right?
MARSHALL: It is kind of a scoop.
WALKER: That’s incredible. Can you tell me a little bit about how that works with the PCR and the genetics?
MARSHALL: PCR is polymerase chain reaction. The coincidence is that Kary Mullis discovered that the same year that I and Robin Warren discovered the importance of Helicobacter pylori in 1983. It was a bit of a landmark as the new biotech started to come in.
So let’s talk about PCR. With all bacteria, all life forms—and criminals, as you know—everybody’s got unique DNA, and bacteria have got a unique DNA.
So with, for example, [Darwins’] whiskers, you dissolve the DNA off them in a test tube and shake it up. Then you put it into a polymerase chain reaction set-up in a machine. If it finds the particular sequence of H. pylori sequence in there, you put a probe in and look for that, it will amplify that little piece of DNA a million times—or a thousand million times. Then you can take a bit of that fluid out of the test tube and put it on a gel, and you can see this little band of proteins appearing. That won’t happen unless you’ve got an exact match.
That’s why PCR is an amazing thing. And, of course, Kary Mullis won the Nobel Prize for that in 1993, I think—ten years later. Sadly, he passed away about two years ago. In the last 100 years, a lot of important Nobels, but that would be one of the best.

WALKER: I was trying to find examples of historical figures who’d suffered from stomach cancer. One of the more famous ones was probably Napoleon. I was reading a good study of his autopsy results, and when they opened his stomach up—he died at the age of 51—it was just a cancerous mess. If stomach cancer was the cause of Napoleon’s death, then you could say there was probably a 90% chance he was infected with H. pylori as well, right?
MARSHALL: Yep, yep. When you finally get the stomach cancer, in the last few years as the cancer is developing, the Helicobacter may disappear, in fact. Once the cancer has started—well, you know it’s going to happen.
The idea is, if you have Helicobacter all your life, when did you catch it? You caught it at two years of age or five years of age from your mother, probably. Then it causes irritation in your stomach, inflammation, all your life.
Fifty years later there’s so much damage in your stomach that you can’t make very much acid, so then the acid gradually declines. Eventually other bacteria start living in your stomach—bacteria from your mouth. The stomach becomes just like your colon; there’s stinky, putrid bacteria living there.
At that point, Helicobacter can’t handle it anymore and disappears. Most people with stomach cancer have got putrid gastric contents for years before the stomach cancer develops and gets them. So that’s the story. People with stomach cancer don’t necessarily get a peptic ulcer because that’s related to acid. Duodenal ulcers, and most peptic ulcers, are acidic, and that wouldn’t be much of a risk factor for stomach cancer. But the final thing when the acid is gone, now it’s ready to get stomach cancer.
WALKER: I have a question on the connection with duodenal ulcers, so I’ll come back to that in a moment. But imagine you could tally up deaths from all the different types of cancer through all of human history. Which one do you think would be responsible for the most deaths? Would it be gastric cancer?
MARSHALL: People didn’t live long enough to get gastric cancer, so it wasn’t really noticed till the 19th or 20th century, I suppose. If the average lifespan is 45, most people with gastric cancer would get it after that, so you wouldn’t notice.
In evolution, if gastric cancer killed you before you had any children, Helicobacter would have been unsuccessful—it would have just died out. But it’s very chronic, and you do pass it on to the next generations. Probably between 2% and 5% of people with Helicobacter would have got gastric cancer. But I’ll be conservative, I’ll say 2%—that would be like Japanese or Japanese Americans, would be good examples.
Years ago, when I was a kid, I remember my grandparents used to get the abbreviated books from Reader’s Digest and the history of the Mayo brothers who started the Mayo Clinic. When they were kids in about 1910, they used to operate on their dog. They would actually do surgery on the animals around the farm, and I know they operated on their pet dog at some point. But when they started the Mayo Clinic in the 1920s and 30s, stomach cancer was the number one cancer in America.
Mayo Clinic would have the data on that, they’d have the records of the patients even, and a lot of gastrointestinal stuff was first documented and studied at the Mayo Clinic. It was a clinic for surgery into GI problems, cancer, and oesophageal disease. You can find some originals and, behind you on the shelf, Proceedings of the Mayo Clinic, 1929–1939 or ’49, I think. I bought that in Pitt Street Bookshop in Melbourne many years ago.
WALKER: Oh, nice.
MARSHALL: If you look through them, you can find original descriptions of various diseases that have been in the medical books for as long as I can remember.
WALKER: That’s cool. It’s interesting that in the first half of the 20th century, in the US and Europe, the leading cause of deaths from cancer was stomach cancer.
MARSHALL: Probably, yep.
WALKER: I think the incidence of stomach cancer was probably more common in the West than it was in Japan at that stage.
MARSHALL: Hmm, yeah. Maybe.
WALKER: But then, since the second half of the 20th century, it’s declined in the US and Europe. What explains that fall?
MARSHALL: Several things. Even in the 1960s you can look at stored blood samples and see who had Helicobacter or not by antibodies in the blood. When they looked at the Kaiser Permanente collection in California, they could see that in 1966, 60% of Americans were infected with Helicobacter—50 or 60%.
So there was still plenty of Helicobacter around the place, but after 1930 there was this decline in stomach cancer in the US. You might have said, “well, Helicobacter’s going away,” but in fact the decline couldn’t be explained by the disappearance of Helicobacter because there was still plenty of it. Going from number one down to number four cancer in that 30–40 year period—it wasn’t just Helicobacter. It might have been that it was refrigeration of food, more fresh vegetables.
That’s really what happened. Everyone started drinking orange juice and got plenty of fresh vegetables. That was good. There are some studies that show that having vitamin C in your stomach inhibits carcinogens. So there’s Helicobacter, and we would say Helicobacter makes everything else worse. Anything you put in your mouth hits your stomach, and if you’ve got a carcinogen in the water supply or bad food or something, then if you’ve got Helicobacter there, that helps set you up to get stomach cancer.
So we say that better diet, fresh fruit and vegetables, refrigeration—that protected a lot of people from stomach cancer.
WALKER: I see, so the H. pylori amplifies other carcinogens because it eats into the mucus?
MARSHALL: That’s correct. If you don’t have H. pylori, you’ll always have stomach acid, and stomach acid destroys carcinogens. They just get consumed—they’re active molecules—so if you’ve got some acid there, they get eaten up or destroyed.
WALKER: I see. It’s also interesting that as there was this fall in the incidence of stomach cancer in the US and Europe, there was a commensurate rise in the incidence of duodenal ulcers.
MARSHALL: That’s right.
WALKER: I read this claim a couple of times when I was reading some papers, and it seemed mysterious to me, but duodenal ulcers and stomach cancer are virtually, though not totally, mutually exclusive.
MARSHALL: I was waiting for you to say that.
WALKER: So if both are fundamentally caused by H. pylori, I don’t understand how one protects against the other. Is it what you were alluding to earlier with stomach acid?
MARSHALL: It’s related to stomach acid, yeah. And you had asked me earlier about omeprazole and acid blockers and things—it’s all wrapped up. That’s why Helicobacter was never discovered, you see, because the main acute disease that young people, working people, would get would be duodenal ulcer. And let’s worry about the important things. Old people get stomach cancer, that’s just another cancer, so no one was focusing too much on that.
Everybody knew that if you had a duodenal ulcer, you never got stomach cancer. Or you never had stomach cancer and a duodenal ulcer at the same time because duodenal ulcer was a high-acid-level disease. Stomach cancer was a low-acid disease, so why would you expect them to be connected?
However, this is Robin Warren’s insight. He’s like, “Barry, let’s not worry about ulcers. Let’s just focus on this inflammation in the stomach. It’s quite a puzzle, and you know I sometimes see it even in people with duodenal ulcer, so what’s going on? Are these bacteria related to it?”
I said, “Okay, let’s study that. That sounds great.”
We wanted to find how bacteria could live in the stomach. So this is our research. This is what you call curiosity-driven research. We’re not trying to find the cause of ulcers, because we could find these bacteria in plenty of people without ulcers.
He said, “Barry, don’t take biopsies from the edge of ulcers—there’s too much healing and inflammation there anyway and it means nothing. Take your biopsy 5cm away from the ulcer,” which we did.
But of course, we counted the ulcers and everything else we could see in the stomach at the same time, but the biopsies were not of ulcer borders. When the data came out, the bacteria were an indicator of ulcer risk, if you like, and it was practically impossible to get a duodenal ulcer if you didn’t have the bacteria.
Obviously, if you ate enough aspirin, you could get an ulcer anywhere and some of them would be in the duodenum—that was another thing we found. And you could get a cancer. If you had cancer of the pancreas, sometimes it could bore a hole through the duodenum and you’d say, “Well, there’s a duodenal ulcer.” So we had the exceptions that proved the rule, if you like, but high 90s was pretty normal.
The other mysterious thing is that they’ve noticed toxin genes in Helicobacter, and if you’ve got the toxin genes in the Helicobacter, you’re more likely to get stomach cancer.
WALKER: Is that the cagA-positive?
MARSHALL: And you’re also more likely to get duodenal ulcer.
WALKER: So the cagA-positive strain, which is the most virulent H. pylori strain, that’s correlated with both duodenal ulcers and stomach cancer…
MARSHALL: Yes. It increases your risk of both, but they’re supposed to be mutually exclusive—so how do you work that out? The answer is that when you catch H. pylori, you’ve got a high acid level, you’re a young person, and ultimately you get a duodenal ulcer. But there’s acid in your stomach which destroys carcinogens and protects you from stomach cancer while you’ve got high acid level.
So the duodenal ulcer, if you like, is a marker for acid in the stomach, and high acid probably. If you eat plenty of protein, you’ll get a higher acid level.
Even though the germ itself has still got the toxin, it’s causing a statistically stronger inflammation. The inflammation means the white cells are going there trying to kill the H. pylori. The H. pylori is not in your body, it’s on your body, if you like. It’s like dermatitis, but it’s dermatitis in the stomach—or it’s like dandruff of your stomach, if you can imagine that. It’ll be a bit annoying, not too bad, but over the years it gradually irritates the stomach.
Now, the white cells are trying to kill the H. pylori, but to get to it they have to separate the cells of the lining of the stomach. Those cells are protecting your body from the acid. If you’ve continually got this process where white cells are burrowing up through and between the cells of the stomach wall to get to the H. pylori and kill them, then it weakens the stomach wall, weakens the acid barrier, and then ultimately—if it’s bad enough—acid bores a hole in and you’ve got an ulcer.
That is why the toxin-producing H. pylori is more dangerous and more likely to cause ulcers. But with acid in the stomach, the ulcers don’t necessarily lead to stomach cancer. Then, as the years go by and acid level falls, and you’ve got things happening in the stomach. At that point there’s not enough acid to create a duodenal ulcer, because that usually means you have to be towards the upper limit—top 25% of the population—to be getting a duodenal ulcer.
So people with duodenal ulcer do have higher acids than other people, but it’s still within the normal range probably. Most of them don’t exceed the normal distribution. Being in the top 25% would be males, and anybody could be there. Eating meat puts you up there. If you have a low-protein diet and you’ve got malnutrition or something, you’ll never get an ulcer because you’re not far enough up the acid curve.
Okay, so then what happens after your “duodenal ulcer”, in quotes, burns itself out? As you get older, the damage, acid went down a bit. You say, “I used to have an ulcer when I was 20. I was in university, I was getting lots of study, pressure, pressure. By the time I got to 30, I didn’t have to worry about it anymore—it seemed to have got better.” You’re talking to a 60-year-old person and that person now hasn’t got enough acid to get significant ulcers but is becoming a cancer risk.
The inflammation’s there, the toxin is there. The inflammation and the toxin—various things damaging the stomach as you get older and leading to cancer. So that cancer is also associated with the toxin. It’s this concept of inflammation for many, many years, even if it’s not a malignant inflammation, can lead to cancer.
That’s been a big eye-opener, and Helicobacter has been the example that got everybody excited about this. It actually applies to three or four other very common human diseases.
WALKER: H. pylori is kind of the textbook example now of that link between inflammation and cancer, right?
MARSHALL: Yep, yep.
WALKER: So, in terms of resolving this paradox of the inverse correlation between duodenal ulcers and gastric cancer—it’s really about the interaction between the H. pylori and the host’s acid levels?
MARSHALL: Acid secretion, yes. It’s been known for years—see, people in China never used to get ulcers, but they would get stomach cancer. So you do have to have a reasonably nutritious diet. You think about it: not enough protein means people with H. pylori don’t make enough acid; they don’t make enough to get an ulcer.
If you study places I’ve been—China, Japan, South America, Brazil—people who studied acid secretion, when that was a pretty common test, would find that in these countries the amount of acid produced was about 50% normal. For you and me, we might produce 28 or 30 milliequivalents per hour if I stimulate your acid—that’s a maximum. Some people do 35, 40. That’s a healthy, Western male, if you like, diet.
In Japan, when they did that study on healthy young males, it was like 17 or 18—most Westerners would produce at least that. Then, 20 years later, when Helicobacter was on the wane in Japan, they repeated it. And in Japan they now had a more nutritious diet, a bit more protein, the acid secretion in Japanese males went up to about 25 or so.
WALKER: Right, interesting.
MARSHALL: That was the 20th-century “ulcer epidemic”, if you like, because duodenal ulcer started to get in the news. “Oh yeah, all those Wall Street guys, these business guys in Chicago, eating big meals and under a lot of stress—they were getting ulcers. So obviously psychosomatic.”
But humans had never been so healthy as they were in the 20th century in the United States, and they were eating plenty of meat. That gives you a higher acid level, and you’ve got H. pylori, so you would get ulcers. There were a lot of people who didn’t have a good enough diet to get ulcers.
The paradox is that when you get a duodenal ulcer it’s actually a sign of good health, because your immune system is very strong and hammering those Helicobacters, boring through the lining of the gut, and then you’ve got an ulcer. People used to say to me, “When you have an ulcer, obviously your immune system’s defective—the Helicobacter is going crazy and you’re losing the battle and you get an ulcer.” But it’s very likely that it’s the white cells fighting the H. pylori that are doing all the damage, releasing superoxide radicals and things they use to kill bacteria.
One of the first patients I ever tested when I was working with Dr Warren—probably 1981—we had some smears of the mucus in the stomach. It had white cells in it and you could see the engulfed bacteria inside the white cell. We had a lot of evidence like that got us a bit excited that, hey, these seem to be pathogenic, your body hates these germs.
WALKER: I just realised, for people wondering what the duodenum is, it’s the first part of the small intestine that connects to the stomach.
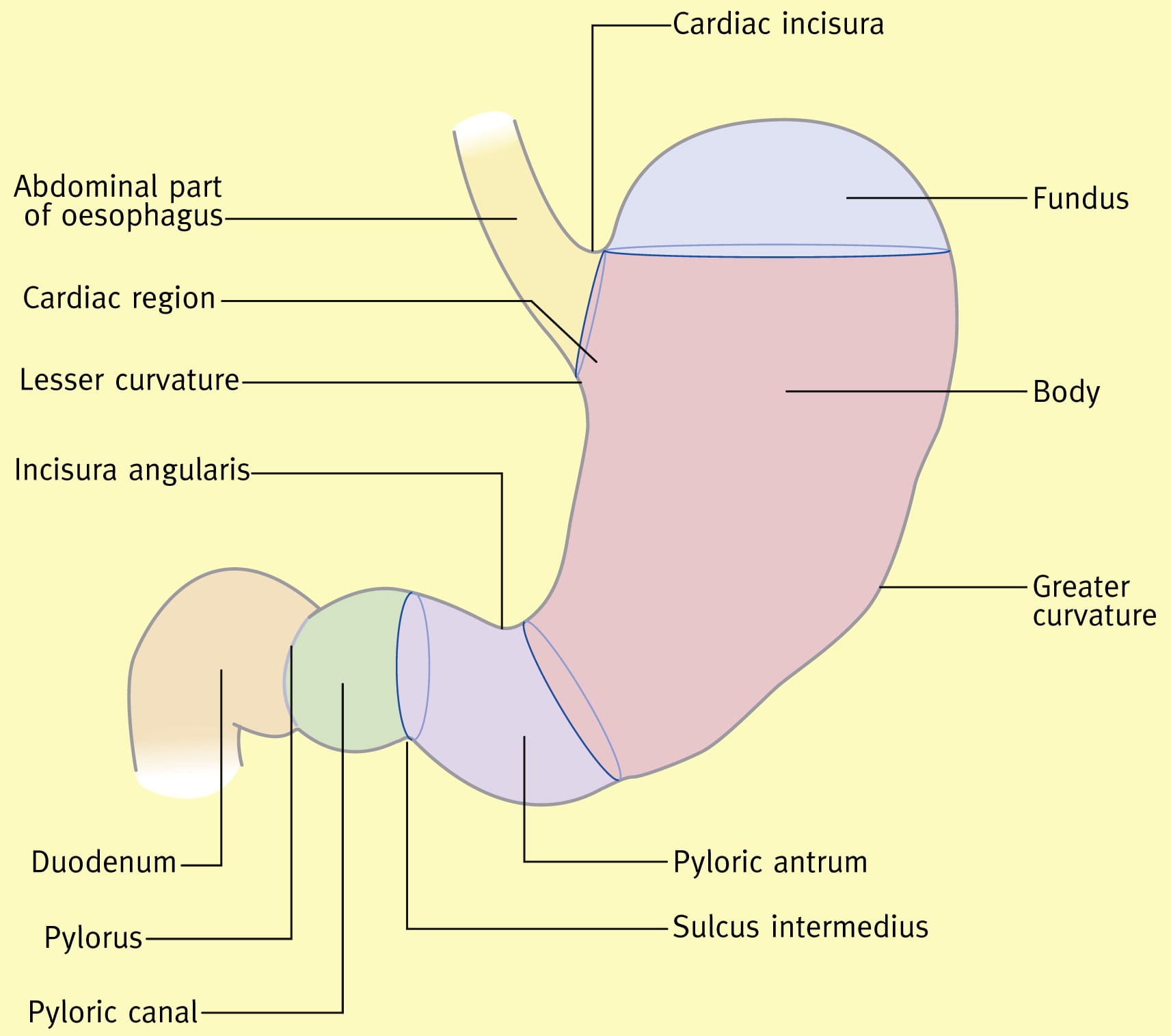
MARSHALL: Yes. The lesson for the stomach is that acid’s incredibly toxic. So it needs to be in the stomach and nowhere else. At the top of your stomach you’ve got your oesophageal sphincter—and if that’s working, that’s great. If it’s not, you’re going to get heartburn, acid reflux, and that’s another illness.
At the bottom end of your stomach you’ve got the pylorus, which is a Greek word meaning “gatekeeper.” The pylorus, I think he might have been the guard at the city gates—they shut the gate at night and, in the meantime, they’d be taxing people coming in and out. That’s why they changed the name to Helicobacter pylori, which is the Greek possessive part of pylorus, I think. Something like that.
So the bottom end is the pylorus. When you’re digesting your food, it’s supposed to stay in your stomach, it gets mixed up until all the food particles are less than about 3 millimetres, and then your stomach will squirt it into the duodenum. The duodenum is right after the stomach, so the pylorus keeps the acid in.
When the acid and food squirts into the duodenum, it’s a little bit toxic. At that point you release bile, which has bicarbonate in it, and the pH of bile is 8.5 or something. Before you allow that toxic acidic mixture to go down into your intestines, in the duodenum it’s mixed with bile and all kinds of juice goes onto it, and it gets the pH neutral.
The other reason you’ve got a stomach with acid in it is to kill bacteria. If you didn’t have something in your stomach to kill all the germs, when it goes into the duodenum and then starts percolating through your intestine over about six or eight hours, bacteria would already be there and be growing. They’d start digesting your sugar and half the nutrients in your food before you actually could digest them yourself. You’d be filling up with gas and feeding this bacterial flora rather than yourself.
Ideally, when the food comes out of your stomach, it then gets neutralised, but it’s pretty much sterile. Then it has four or five hours to get to your colon. By the time it gets to the colon, it’s supposed to have all the good stuff taken out. Then it goes into the colon—it’s a different process, and after that you’re digesting goodies produced by the bacteria.
In gastroenterology, if you have bacterial overgrowth, that means you’ve got bacteria in your small intestine starting from the duodenum down. Then you won’t be able to eat sugar; you’ll be lactose intolerant as well, and all kinds of things can happen to you.
So we’d say you want acid in the right place—not leaking too much up or down when you don’t want it—and you want to have your bacteria in the right place as well, not in your small intestine.
In your small intestine, because you’ve got to absorb food and nutrients, you want that in intimate contact with the actual cells lining the gut. They’ll be taking up the fats and everything and absorbing them. Only when that barrier is broken down, that makes you susceptible to other diseases like cholera and what have you.
So I’d say: don’t get rid of all your acid if you can help it—you know, it’s doing you good.
WALKER: Good life advice. So there’s this set of observations known as the H. pylori enigmas. As you know, the first of these was pointed out by Holcombe in the early 1990s looking at Africa. He observed a very high prevalence of H. pylori infection yet a very low incidence of gastric cancer. Since then, other investigators have pointed out so-called enigmas in other regions like Costa Rica, northern India, and other parts of Asia.
MARSHALL: Yep.
WALKER: What do you make of these enigmas? Are they actual enigmas, or are they illusions?
MARSHALL: It’s a combination of things. You’re right, it probably is correct that the African situation with Helicobacter is interesting—people don’t get as much stomach cancer.
I’d have to say that the data coming out of Africa is not really comprehensive, as you can imagine, and there are plenty of things in Africa that can kill you that are worse than H. pylori. So we’re not too excited about it in Africa.
However, what I can say is that I know people who worked in missionaries and things like the Peace Corps in Africa—in Rwanda and different places. I think it was Rwanda—might have been Uganda—but they reckoned that one in three or one in four African men had a scar on their abdomen where they’d had ulcer surgery. They used to just open you up, do a few cuts in the stomach, and cut the nerves to the stomach so you couldn’t make much acid. That was an antrectomy and vagotomy—those kinds of things. You could end up a bit of a gastrointestinal cripple, not able to digest your food, but a lot of the time that would stop you from bleeding to death from ulcers and put you into remission.
Ulcer surgery was very common. In this hospital in Western Australia—Charles Gardner, where I was—we would do four or five a week, on one surgeon.
So they were very common in Africa, and Helicobacter is not totally harmless—it used to produce a lot of duodenal ulcers.
The other situation: maybe, if people had access to meat—a high-protein diet—in some areas that would help. We didn’t collect much data, and a lot of people never lived long enough to get stomach cancer, because that mostly happens after age 50.
I’m a bit vague as to the reason and people are still wondering about it.
One other answer is that the original Helicobacter did not have the toxin gene.
WALKER: Oh, the one that you and Robin found and cultured—it didn’t…
MARSHALL: No, it probably did. I’m not sure about that—we didn’t have genomics in those days.
WALKER: But is that what you mean by the original?
MARSHALL: No, no—humans have been infected with Helicobacter for 100,000 years.
WALKER: More than 100,000 years ago.
MARSHALL: Yeah—150,000 years ago.
WALKER: Yep, yep.
MARSHALL: And where did humans get it from? They probably came out of the trees, started walking around on the ground, and found a filthy, putrid animal lying there and ate it. It was probably a commensal organism of the ruminants—some kind of antelope that might now be extinct. You don’t know where it came from.
But these bacteria that break down urea and make ammonia are common in the rumen of animals that eat grass. That’s one of the things that helps the digestion of the grass.
Assuming that humans picked it up at that point, so then when you look at Helicobacter in Africa you find these African strains that seem to be ancestral and they don’t have the toxin gene. As you head out of Africa to Europe you get up to 60% with the toxin gene, and then you get to Asia—China is 95–99% or something. So then you look around at different racial groups and migrations, you find some people who seem to have been separated off many years ago and don’t have the toxin gene and potentially would have less inflammation and less disease from Helicobacter.
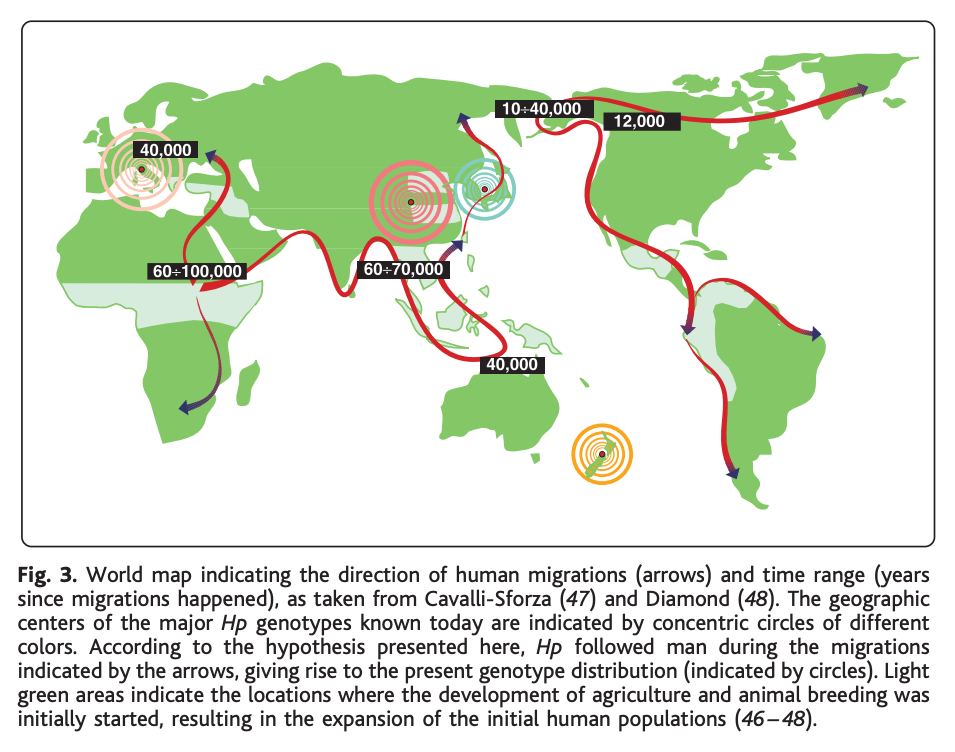
WALKER: It’s super interesting. This is how we can say Helicobacter has probably been travelling with our species for more than 100,000 years. They look at the genetic isolation of different human groups and map that over the H. pylori, and there’s a really good match.
MARSHALL: Yes. That’s right.
WALKER: There’s a well-known model called Correa’s cascade—after Pelayo Correa, with whom it’s most associated—that describes how gastritis progresses to gastric cancer.
MARSHALL: That’s correct.
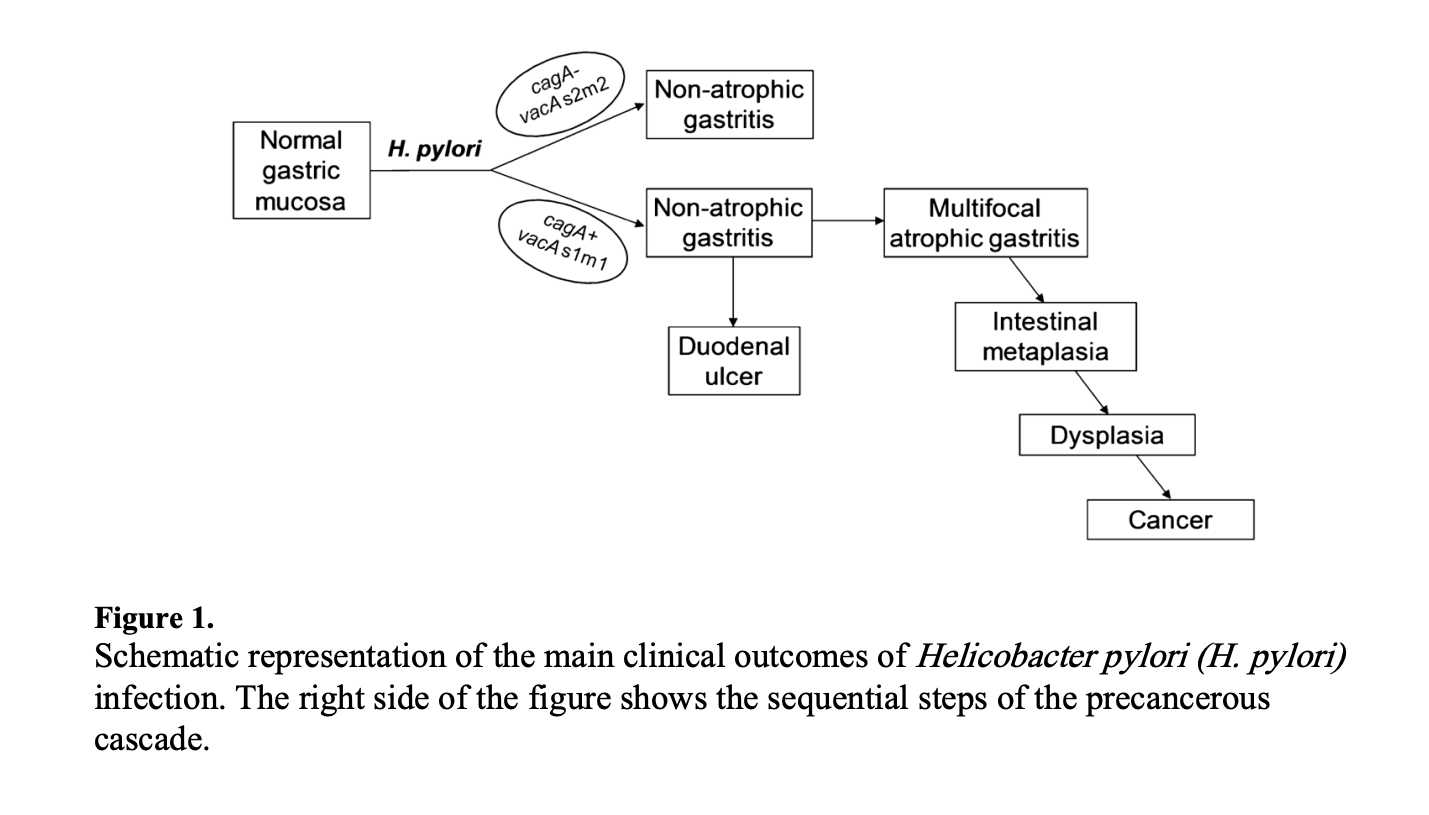
WALKER: You start with normal mucosa in the stomach—that’s the stomach lining. An H. pylori infection can progress to gastritis. Then it can go in a few different directions, but if you’ve got the cagA-positive vacA s1 m1 strain of H. pylori, you can get multifocal atrophic gastritis, which then progresses to intestinal metaplasia, then dysplasia, then gastric cancer.
Question: to what extent is Correa’s cascade reversible? How likely is it that there’s a point of no return beyond which eradicating H. pylori won’t cause you to heal?
MARSHALL: So I think if you’re three-quarters of the way through Correa’s cascade and you stop the H. pylori and the inflammation, most of the time the progression will stop. You’re not going to get more of these islands of metaplasia in the stomach. That’s my opinion, but I couldn’t guarantee that there’s a lot of data from it. These are observations I’ve made over the years looking down the microscopes with Warren...
The effect of Helicobacter pylori on the gastric mucosa is very local. You can see some Helicobacter sitting on some cells and they’re all toxic-looking and sick, and then two or three cell widths away the cells are normal.
If you have a patch of Helicobacter sitting there for a few years it could be damaging that area, and you could have normal cells nearby. One of the problems when you get atrophic gastritis is that Helicobacter damages everything—you get the inflammation—and I can tell you some stories about how inflammation causes cancer; there’s good data on that.
Ultimately the cells that are being damaged turn into intestinal cells. That island is now Correa’s intestinal metaplasia, a little island of it. Once that happens, Helicobacter can’t live on it anymore because it doesn’t stick to intestinal cells. Teleologically, you could say the stomach says, “Oh, Helicobacter’s sticking on the gastric cells—okay, let’s replace them with intestinal cells,” and then Helicobacter is no more a problem. Focally, that is what happens—so it’s an evolutionary thing, I suppose.
Once intestinal metaplasia has formed, although people have tried to show that it goes away when you get rid of the Helicobacter, I’m not sure it does. I don’t see the right experiments being done, because you’ve got 250 little islands of metaplasia around in your stomach.
So when Pelayo Correa looks at a gastrectomy specimen and studies it carefully, he can find these islands of intestinal metaplasia around the stomach. He says, “Oh yes, there’s multifocal atrophic gastritis”—call it MAG, if you like. That’s a risk factor for cancer.
But if you try to study it as a gastroenterologist in live patients, you’re going to go down, “I think it might be there.” You take a biopsy here, you take a biopsy there, and then you come back a year later and try to take another biopsy in the same place to see what’s happened. You might miss the metaplasia. It’s not always obvious which is metaplasia.
You say, “Last year I biopsied here and there was metaplasia, and the biopsy this year doesn’t show any metaplasia. Isn’t that great? It’s gone away since we eradicated the H. pylori.” I’d say, “Well, it may have, but you haven’t proven to me that it’s gone away, because you haven’t taken out the whole stomach and counted it,” or something like he would do in a post-mortem sample. There are some proponents who reckon it gets better.
People say, “Take probiotics.” In China some will say, “Take TCM”—Chinese medicine. But I haven’t seen—well, it’s not proven, if you like.
If I saw a medication on the market in the United States that the FDA had said “yes, gets rid of metaplasia,” I’d be tempted to believe it, because the FDA wants to see data. “Show us people who had 50 biopsies in 500 people, followed for four years,” that kind of thing—million-dollar study. That’s the kind of evidence that would convince people that metaplasia goes away. I don’t think it does.
WALKER: Okay. Yeah, interesting.
MARSHALL: Prove me wrong! [laughing]
WALKER: It’s a challenge to people listening. So, at the very least, around the multifocal atrophic gastritis stage you could reverse that.
MARSHALL: You can stop the gastritis, and it’s not going to progress as much, or at all perhaps. And the data is that you will prevent 50% of future cancers at that point. But you can’t get rid of the total risk of cancer once people have got gastritis and metaplasia.
One thing—I’ll talk about metaplasia briefly—is that they did a study at Harvard about 20 years ago now. They took an animal model of stomach cancer with Helicobacter—cat helicobacters or mouse helicobacters in gerbils. They took female mice and gave them a bone-marrow transplant from a male mouse so they had Y chromosomes in their marrow, and then they gave them the bacteria. They developed stomach cancer, and six months later they could sample the stomach cancer and ask, “What can we find out about these cells that developed into stomach cancer?”
And guess what? They had Y chromosome in them. That means the stomach cancer comes because the inflammation triggers stem cells to migrate into your stomach. Stem cells divide and turn into metaplasia, presumably, and the metaplasia is now the genealogy—the metaplasia is from the stem cells—and that’s what becomes malignant, if you like, because these are dividing cells and they’re susceptible to carcinogens.
You can apply that model to hepatitis B: you have it all your life, you’ve got inflammation—maybe stem cells or something—and you’ve got liver cancer developing. So that’s the cause of liver cancer. Cervical cancer: you’ve got papillomavirus sitting there many years, and maybe stem cells in the cervix lead on to different kinds of metaplasia and cervical cancer. Helicobacter is number one of those three. But it does show that getting rid of these chronic infections, if you live long enough, is going to save your life.
So that’s my understanding of it. There are all kinds of subtleties related to stomach cancer that we don’t understand yet. That’s one of the areas where we’d like to do fundamental research—related to cell biology and things like that.
WALKER: Interesting. At the end I’ll come back with some more questions on infectious diseases and chronic diseases, but can we do the urea breath test now?
MARSHALL: Yes.
WALKER: Okay. I just want to give you some context.
MARSHALL: Okay—yes, I’m wondering about the personal story here.
WALKER: I had never really heard of H. pylori until last year. I’m sure I’d read about it somewhere, and I was aware of your story and the self-experiment where you drank the H. pylori. But I was in the US recording some podcast interviews last year—I was in Berkeley.
I went out for a run one night and just had no fuel in the tank, so I was running much more slowly than my usual pace. I got back, started feeling really nauseous, and then this developed into, I guess, some kind of gastrointestinal sickness over the next maybe five days. The main symptoms were: firstly, nausea; secondly, a feeling of bloatedness, which I’d never experienced before; thirdly, at night, a light fever.
MARSHALL: Interesting.
WALKER: And finally, kind of a strange— I’d describe it as a gnawing feeling in the stomach. It wasn’t pain [laughing], but especially when I hadn’t eaten—a gnawing feeling even after…
MARSHALL: Get up at midnight, open the fridge in the darkness, see the light, and drink the milk.
WALKER: [laughing] Exactly. So I checked into a medical clinic in Berkeley, and the doctor said, “I’m going to get you to do a breath test for H. pylori.” It’s an incredible test—sublimely non-invasive—we’ll demonstrate it in a moment. But anyway, it came back positive. I don’t know whether that was the cause of the symptoms or we just happened to catch it at the same time, but they put me on a quad therapy over there: bismuth subsalicylate, metronidazole, omeprazole, and tetracycline.
After about a week, I started noticing numbness and pins and needles in my fingers.
MARSHALL: Good—good blood level.
WALKER: [laughing] So I figured I was having some kind of side-effect reaction and I stopped taking the quad treatment after the first week. When I got back to Australia at the end of last year, I did two separate negative tests—because I wanted to be extra sure—a few weeks apart, and came back negative.
MARSHALL: Give me the name of that doctor. I’ll write him a letter. Obviously on the ball.
WALKER: Very, very good initiative by that doctor. But I loved doing the breath test because it was so easy, and it’s kind of genius how it works.
MARSHALL: Thank you. [laughing]
WALKER: [laughing] I’ll ask you about it in a moment, but to save you having to explain it, obviously interrupt me if I get any of this wrong. H. pylori feeds on gastric urea. It uses urease to break down the urea into carbon dioxide and ammonia. If you feed the H. pylori, if you ingest urea, you can then measure the proportion of carbon dioxide in your breath to see whether you have H. pylori in your stomach—it’s breaking down that urea.
To be extra accurate, the breath test labels the urea with an isotope—in the case of your PYtest it’s C-14; other tests use, for example, C-13—and that gets tracked through to the carbon dioxide in your breath. So you’re measuring the C-14, and that will tell you whether or not you’re carrying H. pylori. It’s a cool thing. Well, let’s do the test and then I’ll ask you a couple of questions.
MARSHALL: Okay. You have to be fasting—the stomach has to be empty, effectively—so we say fast overnight and miss your breakfast. I know you had a cup of coffee about two hours ago—probably okay. You’ve still got acid in your stomach and there’ll be no other bacteria.
[WALKER holding up test capsule]
The first thing you do is you have the capsule with sugar beads—like hundreds and thousands—and on the surface of those beads you’ve got a coating with a little bit of carbon-14 urea. It’s a beta emitter, so it’s 100% safe, and it’s such a tiny amount that you don’t have to worry about contamination or radiation or anything. You’re going to swallow that with a little drink of water—just enough to get it down.
[WALKER puts capsule in mouth]
Get it down your throat into your stomach, and you start the timer at that point.
[WALKER takes sip of water, swallows capsule]
You want to try to swallow it all down so that—
WALKER: Why don’t you want too much water?
MARSHALL: If you have a lot of water you would dilute the urea. That’s all. Theoretically you might also send it straight down into the duodenum and miss the stomach—the capsule goes surfing down—and you get a low result. You want it to just get in to the stomach. Right now it’s probably sitting at the top of your stomach.
WALKER: Okay.
MARSHALL: And the gelatin capsule is slowly dissolving. Little sugar beads are coming out. Usually, when we looked in the stomach when we were starting this research, we’d find it on the outer aspect of the stomach, about halfway down the greater curve, a little collection of beads about 5 cm in diameter.
Depending on what your stomach’s doing—if it’s just sitting there doing nothing—it won’t spread much. But if you’re feeling a bit hungry and you’re getting a little peristalsis in your stomach, that’s good because it rolls those beads around.
They now have urea on them, which gets released and sits on top of the mucus layer in your stomach. People think the stomach’s like a football, but it’s not—it’s like a wet, empty paper bag. It’s only like a football when you have a big meal. Usually the two walls of the stomach are contracting and moving a little, rubbing together, and the bacteria, and the urea, and the capsule are all rubbing around together.
WALKER: Oh, so that’s why you want to be fasted.
MARSHALL: Yes. It’s getting mixed, and there’s no way that Helicobacter can live there and not be connected with the urea.
[referring to the test] How many minutes have you got?
WALKER: Nearly two minutes.
MARSHALL: Okay. So years ago I was on a plane talking to my boss, going from Brazil to the University of Virginia, saying, “It’s a damn puzzle—how do those bacteria survive in acid?” I had already invented the breath test by then, and I thought, “Oh, that’s why—it’s got urease; it’s making ammonia; it protects from acid.” We went back to the lab, did an experiment to prove this, and got it published in the United States. So it’s proven.
WALKER: Sorry—so the ammonia creates a little force field around the H. pylori. Because ammonia has a pH, it helps to neutralise the stomach acid.
MARSHALL: Yes. H. pylori likes a pH of 6. Or more—6 or 7. So, neutral almost. But the acid is 2—it’s like battery acid you normally have in your stomach. So H. pylori is cranking out this urease. Urea is in your saliva and in your whole body. It gets concentrated and excreted in your urine.
WALKER: In your blood [too].
MARSHALL: It’s at a level of about 2–2.5 millimoles per litre in your blood (per 100 mL).
Helicobacter can survive as low as 1 millimole, and all the other bacteria will be killed in a few minutes—but Helicobacter is still there if there’s urea present.
That’s the experiment we did. So now we know why it’s the only bacterium that lives in your stomach. It doesn’t have to have any competition from other bacteria. It just lives there. And it doesn’t need all these complicated enzymes and special mechanisms that E. coli has—because that lives in the colon and it’s really competitive there. There’s no competition to H. pylori. “So I’m just a dumb little bacteria—just make urease; pretty easy.”
WALKER: It’s got a monopoly on the stomach.
MARSHALL: Yeah.
So it’s drinking time, isn’t it? [referring to the test]
WALKER: Three and a half minutes.
MARSHALL: You’re going to take your second drink of water now.
WALKER: Okay—why do I do this?
MARSHALL: If the capsule was stuck in the oesophagus for some reason—if you hadn’t swallowed it all the way into the stomach—by now the capsule would have fell to bits. This just makes doubly sure the capsule is in the stomach, or the urea has gone in.
WALKER: So the first ever urea breath test was Trimed’s PYtest, right?
MARSHALL: Yes—that was the work I did in the early ’90s at the University of Virginia.
WALKER: Can you tell me the story of coming up with that?
MARSHALL: It started off—I was thinking about Helicobacter as far back as 1984 and I developed a biopsy test based on urease and ammonia production—CLOtest. I was working with that and thought, “It’s a hell of a lot of work for me actually, to do endoscopies on everybody all the time; if only I had a non-invasive test.”
So—serology. We started doing antibody tests. That was great, but not specific enough. I thought, “If I gave you some urea, I wonder if the bacteria would produce enough ammonia to be detected in your breath—or not ammonia in your breath…” We did some studies and found that if you had Helicobacter, you did not have any urea in your gastric juice—it had all been used up. It comes in your saliva and it’s in all your fluids.
But if you had Helicobacter and we sucked out some juice out of the stomach, there was no urea in it—just ammonia.
I found some ancient literature in the chemistry books about how some people had urea in their gastric juice and some had ammonia—you didn’t have both. So I said, “Two and two makes four—obviously these people had urea.” You could look at old chemistry books and see who had H. pylori.
WALKER: It was a clue.
MARSHALL: I then got hold of some carbon-14 urea and I got a protocol at Royal Perth Hospital— so that was ’85. But I worked it out a few months before that. The doctors I worked with then—most are deceased—but Ivor Surveyor was a great guy; he was the head of Nuclear Medicine. His second in command was Professor Agatha van der Schaaf—she’s retired now, I met her recently.
They’re like, “Barry, we’ve looked at the books—it says urease is present in people’s stomachs; everybody has it—waste of time. But all right, we’ll try it out.” So we gave some isotope—carbon-14 urea—which I had by then.
I think the first person to do it was Professor Agatha van der Schaaf. One of those naughty things where we didn’t do an ethics committee, because the ethics committee would have come back and said, “Agatha, should we approve this?” She’d say, “Yes—it’s me; I’m the volunteer.”
WALKER: Self-experiment.
MARSHALL: Sort of a self-experiment. Anyway, she was negative. Then we did get approval. Dr Fox was head of Nuclear Isotopes then and he approved it. We did about 20 people—10 were positive and 10 negative—and it just worked like a dream.
Equating it to the doses we give now—we were giving ten times as much carbon-14 urea in those days, because that’s what they were used to using. Even that’s a tiny dose. Compared with what we do now, the negative people were giving a result of zero or five counts in the breath sample of radioactivity—that’s so close to background. If they took the pill, the C-14 urea was giving counts of about 2,000.
So many times in medicine it’s normal if it’s 3.6, abnormal if it’s 4.6—like a 20% difference; that’s the sort of thing we worry about. To have a diagnostic test where a negative is 10 counts or something and 3,000 counts if it's positive—everybody gets excited. Patients: “My God—3,000!” It was good for doctors and patients—it was pretty much black and white. If you were positive, it was through the roof. And the beautiful thing is, after you got rid of the Helicobacter and went back to normal, it came down to 10, or 0. It motivated the patients: “Those antibiotics are really worth it.”
In those days I used carbon-14 because we didn’t have any carbon-13 technology at Royal Perth—it’s a stable isotope and you had to put it through a mass spec. The technology moved on, and by the time we commercialised the breath tests both were on the market. Carbon-14 was the easiest—and still is the easiest and potentially least expensive.
So for developing countries—and many countries—they embraced carbon-14. Right now in China at least 20 million tests a year would be done using that. I don’t know what it is. Several people do it.
WALKER: [referring to the test] We’re at 9 minutes 30 here.
MARSHALL: Okay. So what’s been happening in your stomach is: the capsule’s spreading around little granules; the urea is reacting—or not. You told me your recent tests were negative, so we’ll probably get a result on you, like 15–20 counts. If we graph your carbon-14 excretion in your breath, it will peak at about 15 minutes. So I don’t care whether we did it at 10 minutes, 15 or 20—it’s going to give the same result.
We chose 10 minutes because that’s as good as the other numbers; if you’re positive, results are already sky-high at five minutes, but occasional outliers might take 10 minutes or so. Ten minutes is a good time—and that’s what we do routinely. If it were 15 minutes it would be the same.
[referring to WALKER’s test] At this point you are going to give me a breath sample. If Helicobacter is there, some of that isotope urea has broken in half and released CO₂. It’s only a tiny bit of CO₂, but it means the isotope will be coming out in your breath. A tiny bit of the CO₂ you breathe out—like a millionth of it—will be an isotope one, and that will give us a positive result.
WALKER: I’ve noticed with other breath tests I’ve done—the ones at the end of last year—they took a baseline sample as well, but you don’t really need that?
MARSHALL: The baseline on carbon-14 is zero—it’s always zero. But the baseline on carbon-13 is 1%. It’s a naturally occurring isotope, and if you eat a lot of corn it might be 1.5%, say. That’s too much noise for the result, so you have to take a baseline. It’s not going to change in an hour from your baseline unless there’s Helicobacter, and it would go from 1.10% up to 1.12%. That tiny difference is what you measure with carbon-13.
WALKER: So that means all the tests I’ve done have been C-13 tests. So this is my first PYtest.
MARSHALL: Yes. With the C-13 test, you’d do at least 20 minutes. Now, move the straw in—there is a self-sealing balloon. Push it down; it’ll slip into the balloon. Push it down. Okay—now wait a minute, I’ll give you some instructions.
[WALKER attaching straw onto balloon]
We want lung air, not mouth air. Take two breaths in and out, then one big breath in—and hold it. Okay, this time breathe in and hold this breath. Hold it—don’t breathe—and we’ll count for 10 seconds.
[MARSHALL counts down from ten to one]
Now blow that breath straight into the balloon until it’s full. Pinch it off at the bottom with your finger and pull the straw out.
[WALKER following the directions]
A little bit leaked out, but that’s still fine.
WALKER: Is that still enough?
MARSHALL: So what you’re going to do is tie that in a knot. A simple granny’s knot. It is self-sealing.
We need a litre and we’ve probably got 1,500 mL there.
When you hold your breath, you’re getting lung air, and we get a good concentration of CO₂—about 5% or so. If you were taking several breaths, a lot of what you breathe out is what you just breathed in and it’s in your trachea or mouth.
From that breath sample we want 1 millimole of CO₂. So we’ll take that breath, we’ll bubble it through an alkali solution—could be sodium hydroxide or similar—and that will suck up the CO₂ until we have 1 millimole.
If we had 2 millimoles in the balloon, the test would only collect one. Then we ask: in that 1 millimole of CO₂, how many counts do we get? At that point everybody’s equivalent—little person or big person. We collect 1 millimole of CO₂ and see how many counts we get per minute.
It’s hard to imagine how many atoms there are in a litre of anything—or a gram of something—it’s like 10²⁰, impossibly big numbers. One in a trillion atoms in your body is carbon-14—you have it naturally. When we give you some carbon-14, we’ve effectively doubled the amount of radioactivity in your body.
It passes out in your breath and urine in 24 hours. That’s why it’s safe. Carbon-14 lasts for 5,000 years, but it’s continually passing through your body. We’re only measuring the occasional ones that liberate an electron—that’s called beta emission. Not like gamma rays—it cannot penetrate your skin or paper. That’s why you need to collect a sample, and every single carbon-14 atom in that balloon is going to be detected by the breath.
So if we’re counting a thousand, that’s an incredibly small amount of carbon-14—because there are millions of them. The technique of counting carbon-14—you know what that was invented for, don’t you? Carbon dating.
If you dig up 10,000-year-old bones, you carbon-date them because they release a tiny bit of carbon-14. Tiny amounts. That’s how sensitive the carbon-14 technology is.
WALKER: Okay, so it’s amazing.
MARSHALL: That’s why, when we put it through the FDA, some people were a bit alarmed about radioactivity. But when you do the calculations, you get down to the fact that it’s background—you get a “breath test” every time you go on a plane. Every hour in a plane you get a breath-test equivalent, and no one worries about it. You get the same amount in a day of background.
The other funny thing is the radioactivity in your body is not only carbon-14—it’s potassium. Potassium in your body releases gamma rays. If you sleep in the same bed as your partner, you’re getting gamma rays from your partner your whole life—that’s about 15 breath tests a year. [laughing] You can’t avoid it.
The average person receives 300 (US units) millirems—that’s like a thousand breath tests a year equivalent, just from normal background. If you live in Denver it’s twice as much—it’s an altitude. So the radioactivity in the breath test is much less than a dental X-ray.
So don’t stress. We don’t worry about children or pregnant women. The FDA had a big meeting about it and signed off on it. There are people still paranoid.
But you can do quite a few tests—you could test the whole family in half an hour; it’s only 10 minutes—you do them all at once. Completely safe, absolutely no side effects. You can send those balloons through the mail—everything.
WALKER: So if I give this to Alfred, will someone actually measure this for me?
MARSHALL: Yep, so Trimed—on your way home, stop in on the way to the airport if you like. They’ll suck the air out of it and run it through the machine, which only takes five minutes or so.
WALKER: We’ll organise that. But for people wondering what my results are, I’ll post them in the comments on the video on my YouTube channel.
MARSHALL: I’ll give a guess—I’ll say 15 counts. And anything less than 50 is negative.
WALKER: Be awkward if I’m positive. [laughing]
MARSHALL: If you’re positive, that’s a strange thing. Are you married? I’d have to find your partner and test the partner—because that’s the only way you could catch it.
WALKER: My girlfriend did a test after I did mine and she was negative.
MARSHALL: Okay, that’s good.
The chance of spreading to your partner over a few years is 50%, as far as I can tell.
WALKER: Okay. So I can have my second coffee now?
MARSHALL: Yeah—let’s go for it. I’ll have coffee as well.
WALKER: Cheers. So, you mentioned the CLOtest, which was the earlier test you developed. “CLO” meaning Campylobacter-like organism—which was the original name for H. pylori until ’89. And the CLOtest was for testing urease in gastric biopsy samples.
I was reading that when Trimed USA eventually bought the manufacturing rights to the CLOtest, you wanted to try to keep the manufacturing in Perth to build up the industry here—but that effort ultimately foundered. I was curious what that experience taught you about either the difficulties of manufacturing generally, or the difficulties of trying to manufacture in Australia—or Perth specifically.
MARSHALL: It was great—we had good support in Western Australia. At that point Technology Park near Curtin University was starting up, and that was a biotech incubator, if you like. A company there—Delta West—mainly made transfusion water and things like that, plus a few creams, but they did have some good equipment and some smart chemists. They actually optimised this test.
It was a little test—a bit of gel. You take a biopsy sample at endoscopy and stick the biopsy in this gel, and if ammonia is produced—it’s like litmus, if you like—it goes from yellow to red. The beauty of that test was the gastroenterologist who believed in H. pylori—at that point nobody believed it, except a few aficionados—could make the diagnosis themselves.
If I was interested in it but my hospital microbiologist didn’t believe it, or my hospital pathologist didn’t believe it, they wouldn’t give me the diagnosis. But if I was a gastroenterologist with this little test, I could put it in my pocket after putting the biopsy in, and look at it 10 minutes later—and if it went red, I’d made the diagnosis very accurately.
The positive predictive value—if the test goes positive in, say, 20 minutes—it’s 100% certain you’ve got the germ. Patients like to see the nice red colour developing—“Oh my God, I’ve got a lot of Helicobacter there.” It helps motivation, and it really took off.
The beauty was you didn’t need a lab or any other people to diagnose and treat Helicobacter. But to see if you were cured I had to do another endoscopy on you—which is a lot of extra manpower. Of course, if I’m a private gastroenterologist, that’s money in the bank—so gastroenterologists thought it was good. But what was your question about… That was the first test, and it was made here…
WALKER: Yeah. So what did it teach you about manufacturing?
MARSHALL: Yeah. So the story about manufacturing and that. I remember I was excited about it and nobody else was, but I showed it to my good friend Rod, who’s been my business manager in Australia since the year dot. I said, “Rod, I reckon we could make this test for 5 cents and probably get 25 cents for it.” He looked at it and said, “Well, Barry, I think you’d probably get $2.50 for it.” [laughing]
Together we then connected with Delta West, and they did the patenting and did that hard work.
After a few years, the issue with this product was it was a refrigerated product. When we shipped it to America, we had a refrigerated box, which effectively doubled the manufacturing price to fly, you know, 100 kilos of these tests. Maybe we’d do 50,000 tests in a box, but it had to be a refrigerated box. That would go out maybe once a month.
If the flight was delayed it might end up in the summer in Chicago on the tarmac and the refrigerator failed or something—and 50,000 tests went up in smoke. They would deteriorate. We used to fly them to Chicago so they arrived at midnight, and they would straightaway get onto a truck. If you drove them down to St Louis—which was our distribution centre; it was Kansas City, I think, actually—you’d drive them down in the night; they’d arrive at 6 o’clock in the morning, it was still nice and cool, and then they’d go into the fridge. That’s how we solved it, but it was a bit of a hassle.
The people who were our distributors in the United States said, “Well, Barry, I’ve got a big company.” Ultimately it was Kimberly-Clark, who makes all the world’s tissues—Kleenex.
The problem was that by then I had a little company that I was a shareholder in that was doing the distribution—doing pretty well. If Delta West sold it to somebody else, I would no longer be connected to the distribution, and so my mates—all that hard work they’d put into releasing it in the US would be lost. So we said we should buy it. My mates in America got some money and bought it and then ultimately resold it to Kimberly-Clark.
Straight away they had the PYtest and the CLOtest, and all the investors got their money back at that point. The West Australian investors got US$7 million for selling a machine that makes them—which is a manufacturing robot built in Perth by a guy, Peter Clark. He often works for the government now—pretty senior—but he’s a government adviser on technology. So, thank you, Peter. He built a good machine that could produce a million of them a month or something, and shipped that over to the United States, and then it was made in the US. That’s where most of the sales were at that point. So that was good.
At that point I actually returned with my family from the US with a bit of a bankroll, paid for my house and all that kind of stuff, and started working with NHMRC funding.
So it got to the US—but it’s not like they snuck it away from us. They paid through the nose to Delta West to get that facility moved to the US. It wasn’t that we couldn’t produce it or create the technology, because they just bought the machine and shipped the machine out.
So whenever I see this story about “poor Aussies—they’ve been rorted again; the new discovery is now being made in tech in the US,” you need to delve a bit deeper and say, “Well, who owned it and what did they pay for it?” And “were Australians prepared to pay that for it?” They were not. To get US$7 million out of Perth investors would be pretty hard.
WALKER: Yeah, that’s not bad.
MARSHALL: I suppose you could have floated it on the stock market maybe—people did that—but there’s a lot of rorting that goes on with that kind of thing and over-hyping, and that also happened with CLOtest over the years—various ownerships and things in Australia. I felt there was a bit of rorting that went on, but I was not party to it. I used to read about how fabulous it was and I’d say, “Wow, did I do that? When did I do that?”
WALKER: [laughing] That’s funny. So it sounds like the main constraint on manufacturing in Perth was just the geographic isolation of Australia. Anytime you need to transport things it causes these huge difficulties.
MARSHALL: Yep.
WALKER: So, one of the many reasons I was very excited to talk with you is I think there are so many meta-scientific lessons from your and Robin’s story. One question I was asking myself was: why wasn’t H. pylori discovered earlier than 1979? I was reading through the book you edited, Helicobacter Pioneers, which is actually an excellent case study in how science works, because you have for about a century, in different places around the world, you have researchers feeling the elephant from different angles.
MARSHALL: Exactly right, exactly right.
WALKER: And you get that in this because each chapter is a different story in the larger story of the discovery.
MARSHALL: I kept meeting people all around the world who told me that their father had discovered H. pylori.
WALKER: It’s funny that the chapter titles are like, “H. pylori was discovered here,” “H. pylori was discovered here,” and you get all or most of the heavy hitters who are still alive to write chapters. I was using this book and trying to…
So there were four clues to the existence of H. pylori which, if you put them all together, you could have deduced the existence of the bug without laying eyes on it like Robin did.
The first clue was endemic hypochlorhydria.
Then there was the existence of gastric urease.
Then there was this strange result that we could treat peptic ulcer disease with antibiotics.
And the fourth clue was that there were spiral bacteria in the stomach—in different mammals, but then by about 1940, we’d seen—
MARSHALL: —we’d forgotten about them. [laughing]
WALKER: —we’d seen them in humans. But I was trying to work out: how big was the window in which all these clues were overlapping?
MARSHALL: Looking back on it, what I would say is that new technologies are continually being invented. They might be invented for problem A—a good example would be cell phones. You need to talk to people when you’re away from home—okay, cell phone. But of course, 90% of the use of a cell phone is for everything else: it’s an alarm, a body monitor, getting information on the Internet, watching movies—all that sort of stuff. When I got my first Motorola flip phone—1993 or something—I said, “This is great; I can make phone calls from my car.” That was about it.
So now that also happened in gastroenterology. And also there are things that happen, like World I and World War II, and scientists are pushed hither and thither—kicked out of the US, for example—and now they’re somewhere else. All these things were happening.
If you look back on it—you talked about antibiotics—well, bismuth, which is one of my patents as well. I discovered that bismuth kills Helicobacter and a lot of other bacteria. That had been used as a component of antacid mixtures for 200 years. People in England were taking it in 1850 for stomach aches. We don’t know what caused the stomach aches.
WALKER: Including Charles Darwin.
MARSHALL: Right—okay—Charles Darwin sometimes was taking bismuth. But it wasn’t scientific; nobody knew that it was good for bacteria. But in fact, it used to be used to treat syphilis—it was painful; you’d get these bismuth injections with black stains on your buttocks.
Bismuth was a recognised antibiotic. But why would you want to use it in the stomach? Why would it be important to the stomach? No—it’s an antacid as well. So that was that one.
The next one was spiral bacteria in the stomachs of animals. That was published in different places—in German literature. If you’d told me that bacteria can’t live in the stomach, I’d say, “Well, acid there, obviously,” but I hadn’t read that German literature from 1890 showing pictures of all these bacteria in dogs’ stomachs.
WALKER: So that was Bizzozero.
MARSHALL: Yeah. The information didn’t flow very easily in those days. They were held up by the different languages. So we talked about the bacteria.
The next one was the urease. Urease had a fabulous history in chemistry. It was the first enzyme that was purified, and it was shown to be a protein. Before that they didn’t know what enzymes were. It was found in the dog’s stomach also, and there was a guy called Murray Luck who did all kinds of urease experiments.
WALKER: Was this when it was discovered in 1925?
MARSHALL: Yeah—Luck and Seth—and they did a book about urease in the dog’s stomach. They said, “Oh, we think it makes ammonia, protects you from acid, because we find all the dogs have got this ammonia thing in their stomach.”
Then Oliver and Fitzgerald wrote a thesis on urease in human stomachs.
WALKER: This is the Irish group.
MARSHALL: Yeah. They said, “Obviously it protects you from acid, and the urease is produced by your cells, and the ammonia protects you from acid.” They even developed an ulcer treatment where they were feeding people urea to generate ammonia and neutralise the acid—like an antacid: “We’ll take urea; you’ve got the urease enzyme.” But of course they would throw up—urea is like chewing tin foil; it’s horrible stuff. It sometimes healed people, but it was toxic to take. That was Oliver Fitzgerald. He ultimately became the president of the British Society of Gastroenterology. He was famous—but that was his thesis.
Then in America these guys—Leber and Lefebvre; actually they were Belgians, I think—studied urea and urease and ammonia in the stomach, and they showed that if you took tetracycline it went away.
WALKER: Which is an antibiotic.
MARSHALL: Yeah. So they said, “Oh, it’s probably caused by bacteria—end of story—blah, blah—let’s go on and do something else.” I actually met Dr Lever years ago—he’s since passed away—and had this discussion with him. A lot of Belgians used to do this: they would stick a tube down your throat and suck the acid out—how much are you making?—so they could do these kinds of experiments, but nobody picked up on this.
Then endoscopy started. The story is, wouldn’t it be great if we could look in the stomach—see if you’ve got an ulcer or something?
Everybody used to have barium meals—you’d swallow the chalky medicine and they’d tip you this way and that—and they’d see a very primitive, low-resolution, black-and-white negative image of your stomach. They could say, “The wall of the stomach looks like it’s got a hole in it—so that’s probably an ulcer, or a duodenal ulcer.” That was the diagnosis of ulcers in those days. If you had less than an ulcer—just gastritis—well, we couldn’t diagnose it very well.
To look in your stomach you used to have to use a straight tube—like a sword swallower. They’d poke this tube down—this is the technology from the ’30s onwards. We used to do that at Sir Charles Gairdner Hospital when I was a thoracics registrar. You’d look down this tube; you’d have a bit of a mirror on the end; you’d move it this way and that. It was pretty difficult to do—the patient would be struggling, people holding his face, holding him down. Anyway—that was that.
When you had flexible endoscopy, everyone says “Hooray!” Now we can just look in the stomach and you don’t have to have anaesthetic—just gag a couple of times—and we’ll look with this fibre-optic gadget and we can see if you’ve got an ulcer. These are technologies invented to find ulcers—but they didn’t realise there are a hundred other things you can do with that technology. Robin Warren’s there: “Why don’t you take a biopsy? We’ll have a look at the inflammation.” And—what do you know?—we found bacteria.
Warren and I were the only gastroenterologists in the world who were taking a biopsy off every single patient we saw. By the time we put the whole thing together, we had 1,000 patients, 2,000 patients, 3,000 patients in our repertoire to test it out. Everyone, when we started talking about it, said, “How can you be so certain? It’s just a new thing and you seem like you really know it. What’s been going on?” Well, we’d been doing it for three years on thousands of people—and treating them with antibiotics.
So we got pretty cocky about it at that point. What I’m saying is there’s a convergence of different technologies that made us realise it. Now, the fourth one that is really fabulous is that—I think it was 1979—Jimmy Carter and the National Library of Medicine put the Index Medicus—that’s all the medical journals every month—on the Internet. So they had Internet—primitive Internet—by 1980. At that point, Robin Warren and I were getting together—1980–81—the Royal Perth Hospital Medical Library got a telex machine, and we could query the international literature every month, looking for things about stomach and bacteria and gastritis and cancer. We’d send off a request, and a day later we’d get a printout with different publications. So we started to see what was going on in the world.
WALKER: So it was MEDLINE.
MARSHALL: MEDLINE—yep.
WALKER: And—sorry—so it would only give you the citation, and then you’d have to find the physical paper?
MARSHALL: We’d get the citation and send, “Give us the abstract.” And if we wanted to see the whole paper, the Australian National Library would have it.
WALKER: Oh, okay.
MARSHALL: They’d photocopy it and send copies. So we were doing Internet research remotely. The delay was two or three weeks before you got anything back. Overnight you might get brief abstracts or just the title. You’d say, “Oh—number 26—give me that,” and the Royal Perth Hospital Library would get it through their connections and send it back, sometimes by fax from America or Switzerland. We started doing proper research at that point.
Eventually—maybe it was Clinton—said, “It’s free.” Previously everyone had a subscription, getting these big telephone books every three months full of citations. You had to trawl through that. Eventually it was online, and then they said it’s free—you still pay your telephone bill—but you could download 100 references if you liked, and you could see text on your little screen by the ’80s. That really helped.
Early on we were just doing it via telex—that’s how Robin Warren and I found all these things in the literature, for Helicobacter Pioneers. You’d say, “I wonder if dogs have it,” and you’d find the report in dogs.
We found several doctors over the years who’d got excited briefly about Helicobacter but couldn’t get the traction to do further research. The surgical PhD in Oxford I spoke to—can’t remember his name—he applied for a research fellowship to study these bacteria they’d seen and was knocked back, so he went off and did something else.
By the time we found it, it was starting to rise from the ashes, if you like—you could see little seedlings of the discovery popping up around the world. If we hadn’t done it, maybe five years later it would’ve been exposed by somebody in England, probably.
WALKER: Yeah, you often see that in the history of science.
MARSHALL: It’s a convergence of these technologies and the Internet pulling all these other bits of information into one place.
WALKER: I see.
MARSHALL: And there are some fantastic examples of stuff that we dug up. One was a post-mortem study in Minnesota where this nerdy pathologist had done post-mortems on traffic accidents. In Minnesota, if you’re driving to your farm and have a traffic accident, you just freeze to death in your car, so when they do the post-mortem you’re frozen solid and then thaw out. It’s like doing a fresh post-mortem on a fresh specimen.
They actually saw all the inflammation and reported it. They reported that whenever they found a duodenal ulcer in a young person, they always found gastritis. They said, “We don’t understand this—the ulcer’s in the duodenum and the gastritis is in the stomach—what’s going on here?”
So when people came along to me and said, “Barry, people with duodenal ulcer don’t have gastritis—the lesion’s in the duodenum; the stomach is normal,” I’d say, “Well, I know something you don’t know: there’s this study from Minnesota, 1952, that people have forgotten about. One hundred per cent of people with duodenal ulcer have gastritis.” I got pretty cocky—and pretty annoying, I suppose, to everybody else.
WALKER: You had the data.
MARSHALL: Yeah. So that’s the answer—get more data. I often get the question: “Dr Marshall, what can I do to get this accepted, or get this drug through the FDA?” I say: just get more data. They’re only interested in data, those people—they’re not human beings at all. If you go broke and commit suicide because you failed, they don’t care.
WALKER: It’s such an interesting case study in how science works. Just to circle back to my earlier question—why didn’t we discover it earlier? Maybe of those four clues, the latest was the presence of gastric urease. Luck discovered it in ’25, but it’s not connected to ulcers until the late ’40s.
MARSHALL: Yeah—and bacteria in the stomach, they proved it in the ’50s.
WALKER: Yeah, exactly. So say from the late ’40s to ’79 is the window where all these clues are co-extensive—about three decades, give or take. What were the constraints operating in that period that prevented scientists from joining the dots? It sounds like—correct me if I’m wrong—the literatures were just so disconnected.
MARSHALL: You didn’t have a unifying hypothesis. You had four or five different, divergent bits of information. And the Librarian of Congress was this guy—his name wasn’t Joseph—his name was Boorstin. Daniel Boorstin, I think. He was the Librarian of the US Congress and wrote a lot of books. He said the delay in discovery—or the reason we don’t make these discoveries—is not ignorance; it’s the illusion of knowledge.
The overriding thing was: everybody knew ulcers were caused by stress. End of story. Take your tranquillisers. I say, in retrospect, if you get pain in the stomach every time you eat a meal—we rely on food to calm us down and make us feel better—if you don’t have that, you could feel pretty stressed. I know I would.
Once you have a unifying hypothesis, you can test it in 20 different ways by looking for information in the literature. By ’85–’86 we had pulled all this literature together to see how it fits. A lot of it was never online. We might have found it in a veterinary book. I found a lot about urease and urea in Ciba-Geigy Scientific Tables—a book of tables: how much sodium is in your blood, how much in your urine, in women and men and children.
One of the things they had was how much urea is in gastric juice.I could look at that andlooking at that data say, “Right, I’m going to make a breath test now, and it’s going to work,” because I could see this. It confirmed what we’d found—maybe I saw the Ciba-Geigy urea data first and then went out and tested patients.
Once you’ve got a unifying hypothesis, you can test it in the literature—just from information. There are quite a few Nobel laureates who’ve won the Nobel by reading other people’s scientific data. Like Kary Mullis—he made the discovery with a thought bubble when he was driving his girlfriend up to the ski resort on the weekend. He was the discoverer of PCR. He discovered it in his brain. And went and did, like, one afternoon of research to test it out.
Warren and I were kind of like that. I said, “Dr Warren—why don’t we try treating this patient with tetracycline?” Okay—so that was our first patient. And then I said, “I’ve been looking at this literature—this guy in England shows that if you take bismuth for your ulcers, the recurrence rate is only about 50%, versus 90% if you’re taking acid blockers. It could be our bacteria there, and bismuth might be an antibiotic.”
So I got some bismuth and tested it—it was the best thing for killing Helicobacter pylori. It was like penicillin. And it was an ulcer treatment already on the market. That accelerated things. That was the eureka moment, in 1983 at Fremantle Hospital.
We grew the Helicobacter. I got hold of some Pepto-Bismol—a pink stomach medicine that’s been on the supermarket shelves for 100 years in America. We made a disc of Pepto-Bismol, put it on this culture of Helicobacter, and put it in the incubator. Monday afternoon—about 4:30—I said, “Better check those culture plates.” There was a disc in the middle of the plate covered in Helicobacter, and the disc with the bismuth had a five-centimetre-diameter zone around it where all the Helicobacter had been killed.
I said, “Right—get me some of this—we’re going to test it on a few patients.” I probably called Robin Warren and spoke to the pathologist: “Look, I want to test this.” Extremely exciting. That was equally as exciting as the self-experiment result.
WALKER: Oh, interesting.
MARSHALL: That happened a few months before I did the self-experiment, but I got a bit cheeky and started publishing.
WALKER: I want to pick up one other interesting lesson in something you said—the new technology of endoscopes. Often in the history of science, technology becomes the platform for new scientific discoveries. Fibre-optic endoscopes didn’t become common until the 1970s, and it sounds like that was a big enabling factor for you and Warren.
MARSHALL: I was seeing endoscopies from ’75 onwards. It was tricky technology—it wasn’t very bright—we’d have a darkened room to look down, and of course we never used to wear gloves. These things would be splashing stomach juice on all the gastroenterologists—who caught Helicobacter. We did studies on gastroenterologists: they had twice as much Helicobacter as any other medical profession.
WALKER: Yeah, that makes sense.
The 1954 study by Palmer—this sort of titan of American gastroenterology—looked at over 1,100 sub-gastric samples and couldn’t find spiral bacteria in any of them. That solidified the consensus that the stomach was sterile and probably reduced enthusiasm for looking for bacteria in the stomach. How did he miss H. pylori with that many subjects?
MARSHALL: Someone in his lab was cheating. That’s the only thing you can think of.
WALKER: Oh, really?
MARSHALL: Palmer was a great pathologist. He used to run Walter Reed Hospital’s pathology department. You can imagine—World War II—there’s all kinds of body tissue turning up in different places, tropical diseases. They were the doyens of pathology in the United States. If they said something, you believed it. Palmer was great.
I met Stone Freedberg when I was in Boston years later—he was in his 90s, about 96, still seeing patients. He was a cardiologist and a general physician, but he published a paper in 1937 or so finding bacteria in the stomach of patients who’d had gastrectomy mostly for ulcers. I think he found it in 46% of his samples and presented it. Then it was World War II; diabetes had just been discovered, or insulin had just been discovered—there was all kinds of fancy stuff going on in medicine. He became a general physician and a cardiologist—so he did okay.
A few years after the war, Palmer and his friends decided to check the bacteria issue. He said to his registrar or research fellow, “Look, there are 1,100 samples in our biopsy collection—go down and have a look, see if any have these bacteria.” He gets a report back a few months later: “We looked at them—we couldn’t see any.” He signs off on the paper—he was the senior author. I don’t know who else was on it, because you just remember him—you remember when famous people make horrible mistakes.
Everybody believed it and never bothered to look. The only way you could miss these bacteria was by not doing proper research—not staining them properly—or just totally faking it. It was inexplicable, but he was deceased by the time I started asking this question.
WALKER: So either someone got lazy and said, look, if we say there's no bacteria that fits with the paradigm, it's a pretty safe thing to say. Or there was some sort of malicious…
MARSHALL: So Palmer might have said to him, look, this is rubbish. Bacteria can't live in the stomach. Go and look at these things and then publish a paper, and then we'll give you your PhD and off you go. So that was it. And I don't think he ever went and looked at them.
So that gets on to fraud in scientific research and how important it can be—not to do it. If he had seen the bacteria and started doing what we did, the literature was available—the tetracycline treatment study, bismuth—he could have done a lot of that. And maybe ulcers and Helicobacter could have been worked out before 1960.
Since 1960, you know, half a million people or a million people a year would have been dying around the world from ulcers. Millions of people were having their stomach removed and never enjoying their food ever again, losing weight and having a horrible life.
And that was because of that screw up in Palmer, in Walter Reed.
WALKER: That's crazy. One of the other interesting lessons from your story is just this theme of how scientific knowledge diffuses. People might think that it's sort of like a light switch where you come up with a new discovery and everyone just instantly updates their knowledge. But it can take years for acceptance of the discovery to roll out.
You could take the NIH consensus conference in 1994 as the date at which H. pylori and its link to gastritis got the stamp of approval. So that's about 10 years after the definitive paper that you and Warren published. I was wondering, would there be some meaningful percentage of GPs and gastroenterologists who even today in 2025 still ascribe ulcers and gastritis to stress?
MARSHALL: Hardly any.
WALKER: Yeah, okay.
MARSHALL: However, if you could walk the street and ask 10 people what causes ulcers, they all say stress.
So it's still in the media. You know, Tom Cruise — his poor E-type Jags are stuck in Rain Man. You know, the E-type Jags couldn't get brought into the country, they didn't have the documents or something. He's there drinking Pepto Bismol because he's got an ulcer. So you still see it.
WALKER: So maybe a few more decades until the punters get “diffused”.
MARSHALL: Yeah.
But the medical book on ulcers used to be 114 pages, and now it's like two pages. So saved all that wasted time. All the med students could go down the beach.
WALKER: So if you hadn't made the discovery until, say, 2015, do you think it still would have taken 10 years to gain acceptance?
MARSHALL: Yep. Well, I suppose with the Internet and me doing a podcast or two about it and everything… and then maybe it helps if you have a commercial product that someone's going to sponsor, that happens a lot faster these days.
But if I discovered something that was out of patent and was just generic, it's a bit harder, because who's going to spend the money? They're never going to get their money back on it. So what I discovered is that the US system, the capitalistic system, actually works very well if you can connect into the people who are going to spend $100 million and promulgate this new treatment.
So on the one hand, if you couldn't get the financing, nothing would happen. It would be very hard. So we had both of those.
In Australia there wasn’t anybody selling anything related to Helicobacter treatment or doing big clinical trials until I did one in Australia. So NHMRC had to fund it and it took a few years, and then it kind of leaked out and diffused out. And then we had to wait until there was a treatment before anybody really had conferences about it and approved it in other countries. So you have to do that work.
So all you can do as a scientist is focus on the data. You may get a patent or two, but I'd say don't let it ruin your life. Let the university do it because you'll get a percentage of it in the end, and the university's not going to give it away for free. They've got professionals to do that.
That was a mistake I made — I tried to do it all myself a little bit and I slowed it down a little bit. But the connection I made was with Procter & Gamble, who made Pepto Bismol, and they realised they had an ulcer product there. So then money started coming out for research to an extent.
And then finally AstraZeneca had omeprazole, which with amoxicillin was really going to cure a lot of it, and it was going to be easy to take and safe. They already had the product. So then in the ’90s they spread the word on it, and finally in 2005 the Nobel Committee didn’t have any scary doubts about it. It was totally proven.
WALKER: Okay. A couple of questions on the concept of self-experimentation, because your self-experiment in 1984, where you drank the H. pylori to try and prove the link between the infection and gastritis, is probably one of the most famous self-experiments in the history of science.
MARSHALL: I lived to tell the tale. [laughing]
WALKER: And I was curious, given your famous self-experiment, do many scientists confide in you about self-experiments of their own?
MARSHALL: Some. And so I think I set a good example in some way in that if somebody is in an early stage and they can plan a reasonable self-experiment, it's not going to be too dangerous. Ethics committees will approve it these days.
It’s likely that the ethics committee at Fremantle Hospital would have approved mine, but things were moving pretty fast for me at that time and I was just getting frustrated that I could not break this barrier: is it commensal or is it a pathogen?
So the pathology data was proof as far as I was concerned. But we didn’t have an animal model, so you couldn’t do that. And most gastroenterologists and normal people — you show them a pathology slide, they don’t actually see the importance of white cells and it doesn’t resonate with them, if you like.
So it was a stumbling block for me and I thought I needed to do it. Can I risk getting it knocked back? Well, no. And can I risk that it’s going to take months before they sign off on it and give me all kinds of hassles with it? So I decided, right, I need to find out, and maybe I’ll do it.
In fact, I had submitted an MD thesis a couple of years before and animal studies were part of it. And the last line was, if I can’t infect animals, I’ll get a human volunteer. So at that point nobody was shocked or anything. So it had kind of passed muster at that level.
WALKER: Yeah. So I read a paper saying that a lot of self-experimentation happens behind the scenes, but it's just not documented because of potential worries about ethics committees.
MARSHALL: I don’t know. If you had a dramatic experiment like mine, you would probably document it. But actually my paper was written in the third person, you know. So, “a volunteer” did this or that.
WALKER: And it wasn’t for a couple of years until you revealed who that was, right?
BARRY MARSHALL: People kind of knew. But these days, if you tried to get that paper published without an ethics committee, the journal wouldn't look at it. They’d be criticised too much.
These days you have to cross the T’s, dot the I’s, as far as the ethics committee is concerned, because there’s a lot of stuff that’s been criticised over the years. There’s also a lot of important good stuff that came out before strict oversight was in place, but it’s a bit too risky for journals to publish now. If they can say, “Oh, there’s an ethics committee, they signed off on it,” then the responsibility is with the ethics committee.
Recently there was a professor in Barcelona trying out new treatments for COVID. I know him peripherally—he was a great microbiologist. But they found out he had been using one ethics committee application for the last ten years across all kinds of research and just putting the same number on things. He was severely criticised. So you can’t do that these days.
Actually, it’s good in some ways because quite often only studies that are successful can get published. So if you get a great result, everyone wants to publish it. If you tested something and it made no difference, it’s still science, but nobody wanted to publish it in a good journal. So over the last 20 years, all the positive results got published, while 10 times as many negative ones, you didn’t see them. So you thought, “Okay, this usually works.”
Take acupuncture, for example. It’s not easily testable. There are dozens or hundreds of papers showing it works, but the ones showing no effect weren’t really published. No one knew about them.
Nowadays, before starting your research, you must show ethics approval and register it in the clinical trials database. Then, if you say treatment X works, reviewers can look in that database of studies and see, “Hang on, there are 30 other papers that have been started but never got published—so we can assume that they didn’t work. So therefore it’s just a lucky fluke with you.” And they won’t publish it.
So it’s better, safer, as it is, but it does delay things a little.
WALKER: One of the basic critiques of self-experiments is that there’s just an N of 1. Do you think that critique is overrated?
MARSHALL: No, I’d say N of 1 is a starting point. But you want some objective evidence that can’t be subjective.
If you said, “Barry, do you think Helicobacter causes bloating?” I’d say, “Oh yeah, I got bloating.” They’d say, “How do you know?” Well, I feel bloated. But what is that? Have you got a photograph? Is your stomach bigger? No—it looked the same. So I didn’t really have any evidence of what I said, what I felt..
That’s the trouble with N of 1. Say I had something that stopped me feeling nervous: echinacea. I’ll give you some kind of therapy which I think is probably bogus. And I said, “When I take this, I feel better.” That’s the N of 1, and it’s so subjective you wouldn’t believe it.
However, if I said I took echinacea, then had an MRI scan, and these funny little wiggly bits in my frontal lobe have all gone straight, just like normal. I’d say, “Well hang on a minute, that’s something interesting, that’s objective data.” With my experiment, Warren could look at the biopsy and say, “No white cells anymore.” Or, “They’ve come, and then gone.” The electron micrographs showed the cells were damaged exactly like in duodenal ulcer patients. Okay, let’s publish. So that’s the N of 1. But you can’t get away with it always.
In retrospect, there was a lot of data I could have collected in that experiment that would have been other objective data, but I didn’t know it was happening until it was happening.
For example, when I was vomiting, why didn’t I catch a glassful and run it through the chemistry department? They would have found so much data in there. But at 6am, when you’re throwing your guts up in the toilet, you don’t really think about science. [laughing]
WALKER: Not thinking about the pH level of the vomitus. [laughing]
MARSHALL: That’s right. Another bit of serendipity was discovering that Helicobacter ruined your stomach acid. During acute infection, you don’t have any acid. So why would you think it causes ulcers? It was quite a paradox.

WALKER: There’s a phenomenon in the history of science: partnerships of two people seem unusually productive. Famous examples include Marie and Pierre Curie, Carl and Gerty Cori, and Watson and Crick, many, many others.
When I model what makes those partnerships effective, the starting point is complementarity. With you and Robin, he brought the pathology and you brought the clinical side. Some of these partnerships feel a lot like marriages.
For example, a couple of years ago I interviewed Daniel Kahneman a few years ago, who won the Nobel Prize in Economic Sciences in 2002 or 2003. He was the Israeli psychologist. He had a famous partnership with Amos Tversky. They pioneered our understanding of biases and heuristics in psychology, which fed into behavioural economics.
When you read about their partnership, it sounds a lot like a marriage. It was platonic but incredibly intense. Whereas reading about you and Warren, it seemed more complicated. Maybe not so much like a marriage. There were the individual letters in 1983, and the 1984 paper sounded like a difficult drafting process—eventually your wives even got involved one night at your house, and they wouldn’t let you leave the room until you’d settled on a final draft, which was then accepted without any changes—which is impressive.
But I wanted to hear in your words how would you describe the character of your collaboration with Dr Warren?
MARSHALL: Well, Warren was pretty obsessional. People would say he was a bit pedantic on focusing on fine details of pathology. But I respected him because he actually taught me the pathology. And although it was a fine detail, I’d say, “I believe it—those cells should not be like that. This one’s definitely abnormal.” This kind of detail.
People who are interested in pathology:, “Is it cancer? Is it not cancer? Move on, Dr Warren. What’s the next case?” That kind of thing. In a busy pathology department, that would be the usual kind of interaction with Dr Warren. “This meeting finishes in two minutes and we’ve got eight more cases, Dr Warren. Could you stop talking about that one; get on to the next three,” or something. That would happen with Robin.
But you could make the same complaint to him every week, it would make no difference to him. It was water off a duck’s back. That was Robin. He was in stone, as far as that was concerned.
So that was good for him as a pathologist, and actually in his life. A lot of people don’t know that he had epilepsy. He had these occasional epileptic attacks, so he needed to be pretty rigid in what he did, otherwise he could fall off the rails. (He did break his hip at one point, falling off his bike.)
So I respected that.
He used to smoke little cigars, and I used to sit down there with him—I was an ex-smoker—and I’d have a cigar with him.
The interaction was that every week we would do a few hours together, looking down the microscope at these cases.
WALKER: Was that side by side at the bench?
MARSHALL: Yes, both looking down the same microscope—we were looking at a two-headed microscope.
You don’t realise what a pathologist does. In the middle of a session, someone would bring him a slide. He’d say, “Hang on, Barry,” look at it in the microscope, then five minutes later the phone would ring from the theatre.
The surgeon says, “What do you think, Robin? Is this cancer?”
He says, “Yes, definitely a sarcoma. Take the leg off.” Click.
So if you’re making decisions like that, you have to be exact and meticulous—if you like, pedantic or obsessional. So that was Robin.
But I’d be there like: “Robin, I’m going to go and treat somebody.” Or, “I’ve got this patient, he’s still waiting in the emergency, and I think we could use the patient, he might feel better.” Or, “Robin, I’ve got to get some samples from cats. Where can I get them?” I had 20 different research projects going on at once.
But Robin was focused on the results of the pathology. I could say, “Right, let’s go and look at cats or dogs.” So I was all over the place, and it was good for him to pull me back and focus. He’d say, “You can’t publish all that.” Our wives said the same thing—“There are 10 papers here, just publish one and focus.”
I’d be writing a paper with 20 different ideas in it. He’d say, “There’s one idea here: germs cause gastritis, and that causes ulcers.”
Eventually we became compatible. I knew what Robin was like and I accepted the deficiencies in my own personality—maybe vice versa.
It was a good partnership.
I like talking to people and studying, so the fact that Robin would run overtime on a lot of these sessions didn’t really worry me too much. But of course, my wife would say: “How come you’re not home at 5:30, Barry?” I’d say, “I had a session with Robin.” So that used to go on.
But you’re right. We were different. I was looking at diverse sources of information, and Robin was focusing on the statistics and the biopsies and the inflammation.
The great story is, in 1983, 1982, we could not do the statistics on gastritis and Helicobacter to show the association, because the p-value was always point zero, zero, zero, zero, zero… five digits. And we wanted to find out exactly.
So then the new Hewlett Packard 11C calculator came on the market, which could do factorial 64. Robin bought one, and we did it. And it was p = 0.00000000001. It was very satisfying.
WALKER: That’s one of the best p-values you’ll find.
MARSHALL: I’ve never seen anything like it.
WALKER: Was there ever a moment the collaboration seemed at risk?
MARSHALL: When we wanted to publish letters to The Lancet, Robin said, “Well, when you walked in the door, Barry, I already had the association with gastritis pretty much worked out. I had enough data and even some drawings of bacteria. I could have published that.”
I said fair enough. But, since I’d been working with him for 18 months at that stage, I said, “Now you want to publish in The Lancet instead of an obscure pathology journal. That is because you’ve got the next chapter, which is the clinical data. Although we are going to publish about gastritis—this is our letters to The Lancet—we’ve got the whole picture here now because we’ve got bacteria and we know that they cause ulcers, even though it’s not present in these letters, and it could cause cancer.”
So we knew that this observation under the microscope of a bit of pathology really was translational into an important disease. So at that point, I put my foot down and said, “Since I came walked in the door of your office, your publication has to stop at that point, and we’ll be joint authors on the second half. But the reason it’ll go in The Lancet is not because it’s a bit of pathology; it’s because it’s an important clinical thing.”
So we then agreed to publish separate letters. If you read Robin’s letter, you can see it’s pathology. If you read my letter, it’s everything else at that point. But I was the one that worked out it was likely to cause stomach cancer, because I’d read the literature on gastritis and cancer. So the last sentence of our first two letters—you weren’t allowed to say it caused anything, because it was too preliminary, but we already knew at that point—we said, “this could also cause gastritis-associated diseases, for example peptic ulcer and gastric cancer.”
That sentence won us the Nobel Prize.
WALKER: It was a little teaser.
MARSHALL: Robin said, “At that point, if we stopped doing research and became motor mechanics, we still would have won it.”
WALKER: [laughing] That’s cool. Earlier we mentioned that H. pylori has been with us for at least 100,000 years. If it has co-evolved with our species, there’s good reason to think there could be benefits to it. What do you think is most likely to be the most important benefit?
MARSHALL: The only one that’s been identified is that it seems to suppress the immune system a little bit, so you’re less likely to get allergies. Kids in countries where everyone’s got H. pylori tend not to get asthma—less of it.
In New York City, kids who had H. pylori had a 45% reduction in their need for allergy medicines—asthma, things like that.
To stay in your body it has to do something to the immune system, suppress it a little bit. If you’re one of these people who’s a bit of a hyper-reactor, if you’ve got H. pylori, you might come down a bit. There’s conflicting data and we can argue about it, but…
I’ve got a company called Ondek that’s doing research continuously on this idea of having on a probiotic related to killed Helicobacter, so that we could take the edge off your immune system and perhaps reduce any tendency to be allergic and get asthma or hay fever—eczema, we don’t know. It might be almost a probiotic, but it’d be killed Helicobacter.
WALKER: In terms of the benefits of infection, what do you make of the seeming inverse correlation between H. pylori infection and oesophageal cancer?
MARSHALL: If you have Helicobacter, in a lot of people, it keeps the acid lower ultimately, at a time when you’re starting to get oesophagitis and hiatus hernias as you get older. So if you have Helicobacter, you might not get the expression of those disorders as much.
It appears that chronic acid in the oesophagus is the risk factor for adenocarcinoma in the oesophagus. On that basis, I can see it could help protect you from acid-induced cancers because the acid is lower.
But the risk on the other side is that Helicobacter itself causing stomach cancer is, people have calculated, about four times the oesophageal benefit. So these days: don’t worry—treat the Helicobacter and take an acid-blocker and you’re sweet.
WALKER: So the net benefit still makes sense to eradicate.
MARSHALL: Yes.
WALKER: How plausible is it that we could eradicate H. pylori globally like we did with smallpox?
MARSHALL: Ninety per cent of it is going to be just hygiene and knowledge. It’s not all that easy to catch Helicobacter—you catch it off your mother—but these days people clean their teeth every morning and they don’t chew up the food and feed it to the babies like everybody used to do in the past century.
So it’s not super easy to catch it. You’ve got to be in pretty intimate contact with a family member usually, or have dirty water—faeces contamination of your water—that’d be the thing.
As the standard of living rises and understanding of infectious disease spreads, Helicobacter is having a hard time keeping on.
Ultimately, I think there’ll be a vaccine. I’ve connected with groups around the world working on vaccines for Helicobacter.
It’s not easy enough yet and not certain enough to make it worthwhile, but there are vaccines—or at least one. It’d be a traveller’s vaccine: you could take it and it would cut down your risk of catching it if you were in Africa or South America or somewhere.
WALKER: What seems to you like the most promising delivery platform?
MARSHALL: I’d say it has to be oral.
WALKER: Oral recombinant?
MARSHALL: Yeah.
WALKER: Because then it would go into the stomach.
MARSHALL: That’s right. You need your immune system carrying out surveillance in your gut and making antibodies that stick to the proteins on H. pylori and stop it attaching to your stomach. That would be the thing.
WALKER: When you do predict we’ll have an H. pylori vaccine?
MARSHALL: To roll out everywhere, I’d say 15 or 20 years. We actually might have it now, but of course we have to test it in animals and thousands of people. And then, how long do you have to have it before your Helicobacter goes away or you get protected?
As soon as you have a trial, knowledge about Helicobacter goes up in the community and they stop catching it from general public health measures.
WALKER: Why haven’t Western countries tried to eradicate it already?
MARSHALL: With vaccine, or just eradicate it?
WALKER: Just eradicate it through the quad or triple therapy.
MARSHALL: It is a bit more difficult than normal infections. You have to do a preliminary test and then a follow-up test as well. With a urinary-tract infection I’d say, “Just dipstick your urine,” or something—we’re not quite there with H. pylori.
There was always an argument as to whether or not it’s good or bad. People who didn’t feel like treating it would say, “Oh, it’s probably good for you in some way.” In evolution, all humans had it, so you could say it has a benefit. What we think is that any benefit is in childhood, related to allergies—that’d be the thing. After that, you’ve lived too long—time for you to go—and Helicobacter will help you on your way. [laughing]
WALKER: Is Japan still the country to beat when it comes to screening and eradication programs?
MARSHALL: Probably. I think children in Japan are only about 2% infected, as far as I’ve seen. That means the next generation’s not going to have it. And if you’ve been to Japan, you know they are extremely clean. Anyone who eats raw fish all the time needs to make sure there are no germs around.
WALKER: Is there something they do really well in screening and eradication that Western countries like Australia could easily copy?
MARSHALL: No. They just test for it. They have numerous tests on the market and they screen. They’ve had a faeces test, urine test, breath tests, and endoscopy. They already had universal endoscopic cancer screening—and I think they still have it. From age 40 onwards you can have an endoscopy every two years and they’ll take some biopsies and look for it.
So they already had awareness of stomach cancer and protection, and half the population used to do endoscopy. They probably still do. Helicobacter got caught up in that—so half the people already had a way of finding it and treating it, so it happened pretty quickly. Once the government signed off on the treatment and started paying for it, everybody wanted it.
WALKER: Last question, Barry. One way of describing the paradigm shift you and Dr Warren initiated is that you blurred the long-held distinction between infectious diseases and chronic diseases—because we discovered what we thought was a chronic disease was actually caused by a pathogen.
Are there other chronic conditions—arthritis, Alzheimer’s, IBS, schizophrenia, diabetes, obesity—that you think we’ll eventually discover are actually caused by microbes?
MARSHALL: That’s a pretty good list. I’m interested in all of those. Since PCR came out, for any kind of chronic disease people have been looking for germs and viruses using molecular probes and things like that.
Recently, one of these probably bogus papers was finding genomes of bacteria and viruses in bits of brain and in bits of tumours—things like that. It’s a situation of early reports and positive results get reported and everyone’s excited and hyped up. That’s been going on a lot, but pretty lean pickings for most of it. I would say almost certainly some types of Alzheimer’s are probably a viral illness—whether it’s chronic, or damage was done years ago and something leads on.
For instance, polio. I was talking about this the other day: people who had polio as children are more likely to get motor neurone disease as adults than people who never had polio. Nevertheless, most motor neurone disease is polio, is not related to that. That’s the kind of evidence that’s exciting. A lot of autoimmune diseases—like Crohn’s disease and colitis—look like infections. If Warren had seen bacteria on colitis, he’d say those white cells are doing something—fighting the bacteria—but it’s not obvious at the moment.
I say keep looking, but we have to look in early life. My thought is that from the day they’re born, we should be looking at children’s microbiomes and documenting: they had a cold, a runny nose, diarrhoea, vomiting—this, that. Each time something happens, collect a bit and put it in the fridge.
Twenty years later, someone discovers a virus, we get a probe for it, and we look at all those stored samples. That kind of data and biobanking is going to unlock a few of these. (I don’t know how many.)
There’s tantalising data out there. For example, nuns with Alzheimer’s—you could look at their application letter when they were 18 years old to get into the nunnery, in a study in Ireland. You could get a linguist to look at the language and say, “This person’s less intelligent than that one; this person’s not as good at writing letters as that one.” The ones who didn’t write a great letter got Alzheimer’s more.
The ability to look longitudinally at illnesses and at normal people—and notice aberrations or trends—is going to be important. That’ll give us clues. All this big-data stuff and the different kinds of data we’re collecting about ourselves—we’ll all have monitors on ourselves. All my labs—my haemoglobin and everything I’ve had taken for the last 20 years—I’ve got on my computer.
It might not be easy to hand-grab it all at the moment. The health record—privacy is something we should think about, maybe have a privacy ombudsman—but it’s not front and centre for most people; they don’t really care too much about it, mostly. That’s going to be an advantage. Five or ten years from now things will be coming out of it—and they already are. In San Francisco, for example, there’s biobanking: Kaiser Permanente serum since 1960 or so. Every 20 years—or every 10 years—they do a massive randomised study around the United States and collect blood and data from everybody, and it’s in a public database. That kind of thing is going to find a few causes of these diseases.
We’re getting new biologic therapies, working out what autoimmune disease is triggered by—hopefully. We can say something’s an autoimmune disease; we know you’re reacting, but at the moment we can’t tell what started it. This idea of finding the 3D structure of every antibody floating around in your blood and looking at everything else in the environment, we might be able to tell it’s really coming from dandelions or whatever.
I’m very optimistic. But it’s going to take skilled, smart PhDs looking at a lot of numbers and graphs on the screen.
We’re really into the biologic century, if you like. All that information will eventually be obvious to us, just as some information became obvious to me once we could query online databases.
WALKER: Absolutely. Well, I’ve got through almost all of my questions. It’s been really fun talking with you. Thanks so much.
MARSHALL: Thank you for obviously spending a lot of time preparing the questions. It was fun answering because they were slightly different questions to what I usually get.
WALKER: Great. Alright, thanks so much, Barry. Appreciate it.
MARSHALL: Thank you very much, Joe.
